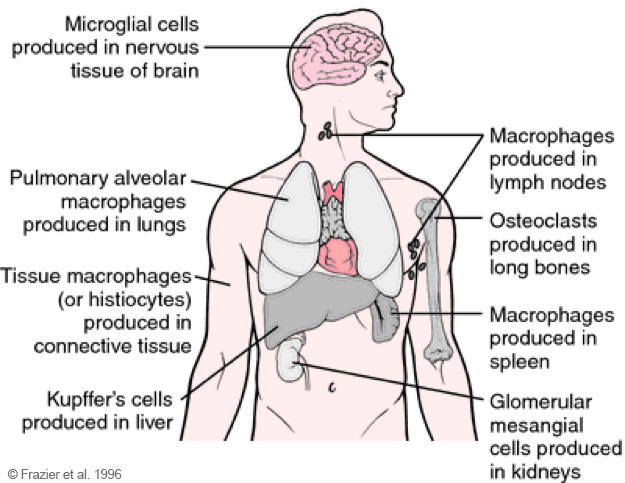
Nanoparticles that are present in the body (i.e. after injection) are largely taken up and eliminated by the so called reticulohistocytic system (RHS). The function of the RHS is the inactivation and elimination of dead cells, bacteria, viruses, and infiltrated small particulates. Nanoparticles also belong to these “infiltrates”.
The reticulohistocytic system is also named reticuloendothelial system (RES) or mononuclear phagocyte system (MPS). It is a network of cells and tissues that are distributed over the whole body and are especially abundant in the blood, liver, spleen, lungs, lymph nodes, bone marrow, but also in connective tissues and the brain. The cells of the RHS take up particular materials in the blood and in the tissues by phagocytosis and degrade and inactivate these materials with the help of enzymes. This network of cells consequently possesses important functions of the immune defences against diseases.
Particulate materials that are taken up by this system include dead cells, bacteria, viruses, fat particles, and solid particles such as nanoparticles. Nanoparticles that are injected into the blood through the veins for diagnostic or therapeutic purposes (for instance for cancer therapy) are taken up in amounts of 60 – 90 % by the Kupffer cells of the liver and of 2 – 20 % by the spleen. The rest is distributed into the other cells of the reticuloendothelial system [1-4]. The body distribution thereby is governed by the surface properties of the particles. For this reason, the blood circulation time can be prolonged or their body distribution can be altered using special coating materials. As a result enhanced concentrations in certain tumours can be achieved [5-7].
Intramuscular or subcutaneous injection lead to a preferential accumulation of the particles in the local lymph nodes [8,9].
Conclusions: Without special surface alteration more than 90 % of the nanoparticles are recognised by the RES, taken up by these cells, and then disposed and eliminated from the body. For these reasons, particles for therapeutic purposes have to be especially designed for instance to achieve an enhanced tumour concentration.
- Scheffel, U et al. (1972), J Nucl Med 13: 498-503.
- Illum, L et al. (1986), Int J Pharm 29: 53-65.
- Illum, L et al. (1987), Life Sci 40: 367-374.
- Tröster, SD et al. (1990), Int J Pharm 61: 85-100.
- Beck, P et al. (1993), J Microencapsul 10: 101-114.
- Reszka, R et al. (1997), J Pharmacol Exp Ther 280: 232-237.
- Lode, J et al. (2001), Pharm Res 18: 1613-1619.
- Kreuter, J et al. (1983), J Pharm Sci 72: 1146-1149.
- Maincent, P et al. (1992), Pharm Res 9: 1534-1539.
 >
>