Toners are very fine powders mainly used in copy machines and laser printers. They are often composed of particles with 2-30 microns in diameter. The small particle sizes cause the powders to behave like liquids. Consumers can come into contact with toner particles or paper dust during maintenance work in the resting state of the device e.g. when toner cartridges are replaced or a paper jam has to be removed. During the printing operation with closed housings volatile substances and nanoparticles can be released through the ventilation system of the device into the ambient air leading possibly to inhalative exposure.
Toner – the Material

Schematic structure of toner particles. © IUTA, 2014.
One-component toners are composed of toner particles that use a polymer resin as base material, pigments (usually carbon black) and magnetisable metal oxides (e.g. iron oxide). The pigments may be incorporated into the base material and/or form a coating on the surface of the toner particles. Additives like silicon dioxide particles are used to prevent clumping and materials such as manganese, chromium or zinc function as coloured pigments [1].
The printing process
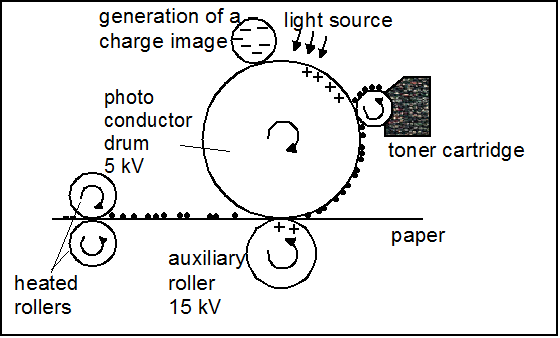
principle of the printing process. © IUTA, 2014.
During the printing the toner is subjected to various processing steps with different release potentials. The principle of the printing process is shown here schematically.
At first the printing template is projected by electrostatic charges on a photosensitive drum. The toner is provided in a cartridge and applied to the charged photoconductor drum where the toner particles adhere by means of electrostatic attraction. To transfer the toner particles to the paper a higher charged auxiliary roller is used. The paper passes between the photoconductor drum and the auxiliary roller with the toner particles being transferred onto the paper by means of electrostatic forces. On the paper, the toner particles are fixed by heated rollers (130-210°C) in the fuser unit [2].
The release
There exist different release scenarios for toner particles than in turn have different influences on the possible impact on human health. Skin contact with the toner particles (2-30 micrometers) or paper dust with subsequent skin irritation can take place e.g. during maintenance work in the resting state of the device for example when toner cartridges are being replaced or a jam has to be eliminated. This contact can be avoided by wearing gloves.
During the printing operation with closed housing, volatile gaseous compounds (e.g. xylene) and nanoparticles may be released through the ventilation system into ambient air. Due to the size of the particles, they can reach the lower respiratory tract when inhaled. The release during operation is mainly caused by the fixing of the toner particles to the paper with the heated rollers. Volatile substances can evaporate from the plastic parts of the printer, the paper and the toner particles which partially condense in cooler areas and possibly forming new nano-sized particles. Analyses of the aerosols from laser printing systems determined silicon, sulphur, chlorine, calcium, titanium, chromium, iron, bromine and traces of nickel as constituents. Iron and silicon are attributed as source to the toner, calcium and chlorine to the paper and bromine to the flame retardants in the plastic housing parts [1,2,3,4,5,6].
Generally, two types of studies investigating emissions from printers and copy machines can be distinguished, the ones that are carried out in real offices during normal office activity, and tests of individual laser printers in test chambers with defined dimensions. These studies show that during the operation of laser printers or photocopiers particles and gaseous pollutants can be released into the air The results showed that particle number concentrations from laser printers can be affected by many factors, e.g. the device type and age, the age of the toner cartridges, the operating state of the printer and its installation location[2,5,7,8,9].

Toner powder in different colours. © Diegofamu / Fotolia.com.
Health Aspects
In vitro studies showed that toner dust in high doses can damage cells. In experiments with rats that were exposed to high doses of toner dust, the particles caused inflammation in the lungs. However, the administered amounts were so high that they do not reflect reality.
Individual studies show that people can react hypersensitive to toner dust. The result of a study with volunteers staying for one day in a copy shop with a subsequent measurement of the inflammatory markers of the upper airways was that the induced inflammations disappeared latest after one day[10,11,12,13].
In principle:
Photocopiers and printers can cause different types of gaseous and particulate emissions such as ozone, UV light, noise, paper-, house- and toner dust, volatile organic compounds and heat. The extent of the emissions depends on the type of device and the circumstances of use (frequency of use, ambient conditions, etc.). Therefore, the potential specific health hazards of printing devices have to be assessed on a case by case basis.
The German Federal Institute for Occupational Safety and Health (BAuA) and the Swiss Suva published recommendations for safe handling of toner dust:
- https://www.baua.de/EN/Topics/Work-design/Hazardous-substances/Working-with-hazardous-substances/Information-on-substances/Toner-dust.html
- https://www.suva.ch/download/factsheets/gesundheitsgefaehrdung-durch-laserdrucker–kopiergeraete-und-toner/standard-variante (in GERMAN only)
Literature
- Barthel, M et al. (2011), Environ Sci Technol 45(18):7819–7825.
- Wang, ZM et al. (2011), Aerosol Sci Technol 45(9):1060-1068.
- Gminski, R et al. (2006), Umweltmedizin in Forschung und Praxis 11(5):269-300.
- He, C et al. (2010), J Aerosol Sci, 41(6): 523-530.
- Morawska, L et al. (2009), Environ Sci Technol, 43(4): 1015-1022.
- Wensing, M et al. (2008), Sci Total Environ, 407(1): 418-427.
- Fiedler, J et al. (2009), Gefahrstoffe – Reinhaltung der Luft, 69(03): 77-82.
- He, C et al. (2007), Environ Sci Technol, 41(17): 6039-6045.
- Tang, T et al. (2012a), Environ Sci Pollut Res Int, 19(9): 3840-3849.
- Tang, T et al. (2012),Environ Mol Mutagen, 53(2): 125-135.
- Konczol, M et al. (2013), Toxicol Lett, 216(2-3): 171-180.
- Khatri, M et al. (2013), Nanotoxicology, 7(5): 1014-1027.
- Morimoto, Y et al. (2013), Inhal Toxicol, 25(12): 679-690.
 >
>