Iron is further processed into steel or electronic data storage media. Iron compounds are also used in catalytic processes in the chemical industry. Furthermore, scientists are currently researching the use of iron oxide particles in medicine, e.g. as a contrast agent or for tumour therapy. Iron is one of the most abundant metals on earth. Its oxides are mined as iron ore.

Train cemetery © Uyuni Bolivia c javarman / fotolia.com
How can I come into contact with this material?
There is little chance for the user to come into contact with iron and iron oxide nanoparticles used in electronic devices and in data storage media. In the medical field, however, iron oxide nanoparticles are injected directly into the human body. Therefore it is extremely important to ensure that the used concentrations of the iron oxide preparations are non-toxic.
Is there any risk from this material to humans and the environment?
The administered amounts of iron and iron oxide used in medical applications are very small compared to the natural iron supplies within the body and are considered non-toxic. Naturally occurring iron oxides are ubiquitously found in the environment making it difficult to distinguish between the naturally occurring forms and synthetically produced iron oxide nanoparticles. Environmental engineers are using this material for environmental remediation purposes such as the removal of toxic compounds from ground water where the nanoparticles usually remain after the chemicals have been eliminated. In general iron and iron oxide nanoparticles are considered to be non-toxic to the environment and its inhabitants, only exceptionally high concentrations can be problematic. Nevertheless it is essential to differentiate between the effects of metallic iron and the respective iron oxides as the former cannot be taken up by organisms. Iron oxides however are important trace elements which can cause adverse effects in both humans and wildlife when administered in excessive amounts.
Conclusion
In the day-to-day life the human body is only exposed to very small amounts of iron nanoparticles or iron oxide nanoparticles which are considered generally to be non-toxic. Iron and iron oxide are naturally occurring materials in the environment.
By the way…
- Iron oxide is also used as colour pigment to create the typical rust red colour. It is a long-lasting dye that can been found in many ancient paintings.
- Pigeons and bacteria are known to store iron oxide particles within their bodies to enable orientation with regard to the earth’s magnetic field.
Properties and Applications

Hard disk drive with open housing © fraeuleinlux / fotolia.com
Ferrous metal and (certain) iron oxides are magnetic, in the past they were used e.g. on audio tapes and on cassettes to store sound recordings, today this is only a niche area in audio engineering. Later on, such iron compounds were also used on (floppy) disks until they, in turn, were almost completely replaced by CDs, DVDs, and storage sticks. However, magnetic recording media is still used in hard drives of computers. With the increased emergence of PCs as home computers and media servers, whose storage capacities are provided by hard drives, one could speak of a renaissance of magnetic recording for the personal music collection.
Apart from iron, cobalt and nickel are also used for modern hard drives [3] that use the Giant Magneto Resistance Effect (GMR [4]) as a base for data recording. Structures of just a few nanometers in size are used here to store the data and it is only these extremely small structures that make it possible today (in 2009) to store data in the range of terabytes on hard drives. Iron oxide is also used as a colour pigment with the typical rust-red colour. Because of its high chemical stability iron oxide is extremely durable as a dye; it can already be seen in antique paintings.
But iron oxide can be undesired as well: oxidation of iron makes for the biggest problem in dealing with this material, as this kind of oxidation is known in everyday life as “rusting”. This process costs the world economy billions each year, because when it rusts, iron looses its mechanical stability and crumbles to small pieces of rust.
Iron is prevalent in the living nature as well, as part of the haemoglobin of our red blood cells it plays as important a role for higher organisms as in the enzyme nitrogenise that accomplishes self-fertilization in some plants. Without iron humans and plants would not be viable. Patients with the so-called “anaemia” are treated with doses of iron salt to support the formation of haemoglobin.
Some newer diagnostic procedures most of them still in a test phase, use nano iron oxide as a contrast medium for medical imaging processes. Such magnetic processes shall - whenever possible - reduce the radiation exposure in medical examinations. Specially coated Fe oxide nanoparticles promise progress in cancer therapy. (Hyperthermia) .
Metallic nano-iron is self-inflammable. The mixture of nano-iron with air (as dust) is also ignitable without the influence of an ignition source. Iron oxide, on the other hand, is not self-inflammable as a nanometer-sized powder. Even as a mixture with air (dust) under the influence of an ignition source, the mixture is not flammable, so there is no possibility of dust explosion in the case of iron oxide.
Natual Occurrence and Production
Iron (chemical symbol Fe, Latin: ferrum) is one of the most important metals in everyday use. Steel girders for bridges and houses, rolled plates made of iron and steel, i.e. for the construction of automobiles, screws and nails - all these consist of iron or of steel, the refined form of iron.Iron is one of the most abundant metals on earth. It is mined as iron ore in the form of iron oxides. Chemically, these iron oxides are Fe2O3, Fe3O4 or very rarely FeO. In 2000 about half a billion tons of iron were manufactured from iron ore and this number is increasing.
First the mined iron ore has to be extracted from its oxidic form to the elemental, the metallic iron. For this purpose there exist huge blast-furnaces [2] that reduce iron oxide chemically to iron, in most cases with the help of coal. Depending on its intended use the iron is then processed to structural or tool steel by adding more metals or by reducing the carbon content.
Literature
- Wikipedia (EN): Iron (last access date: Dec 2017).
- Wikipedia (EN): Blast furnace (last access date: Dec 2017).
- IBM Research - Simulation of an MR and GMR (www.research.ibm.com)
- Peter Gruenberg Institute (PGI), Research Centre Juelich
Studies have shown that so-called superparamagnetic iron oxide nanoparticles are very well tolerated by cells and cause hardly any negative effects. Since iron oxide particles are mainly used medically (e.g. as contrast agents, for tumour therapy), it is important that they do not cause toxicity at the doses used.
Studies on Living Organisms – in vivo
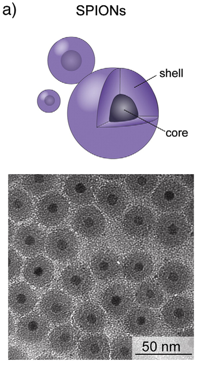
SPION: iron oxide core and hygroscopic shell. © Kunzman et al., 2011.
The SPION currently used are made up of two components: A core of iron oxide and a shell of a hygroscopic polymer The shell prevents the nanoparticles from agglomeration, reduces their toxicity, and controls their behaviour and distribution in the body. SPION particles with dextran shells have already been studied intensively and have been admitted for clinical applications by the US Food and Drug Administration (FDA).
Intravenously injected SPION are mainly taken up by macrophages. In these cells, the polymer coating is degraded and is then excreted via the urine or the stool. The iron contained in the SPION is built into the body’s iron stores. The low toxicity of SPION is not surprising considering that natural iron concentrations in the body may be as high as approximately 4000 mg/adult person while no more than 50 – 200 mg/person are administered during medical treatment.
SPION that are provided with a special polymer shell (polyethylene glycol – PEG) are taken up less readily by the macrophages. The retention time of SPION in the body is prolonged through such modification, and the probability of reaching the desired target (for example, the tumor) is increased.
Literature
- Mueller, K et al. (2007), Biomaterials, 28(9): 1629-1642.
- Kunzmann, A et al. (2011), Biochemica et Biophysica Acta (BBA)-General Subjects, 1810(3): 361-373.
Studies Outside of Organisms - in vitro
Besides an increased production of inflammatory factors observed in some studies, the addition of iron oxide nanoparticles to macrophages does not exert any major negative impacts.
It was found that the toxicity of SPION can often be reduced considerably choosing a suitable coating polymer that covers the SPION surface. Coating polymers may consist of various different chemical compounds. The toxicity of the SPION is mainly determined by the chemical composition of the respective coatings.
Literature
- Mueller, K et al. (2007), Biomaterials, 28(9): 1629-1642.
- Hsiao, JK et al. (2008), NMR Biomed, 21(8): 820-829.
- Siglienti, I et al. (2006), J Neuroimmunol, 173(1-2): 166-173.
- Buyukhatipoglu, K et al. (2009), J Nanosci Nanotechnol, 9(12): 6834-6843.
- Choi, JY et al. (2010), Bioprocess Biosyst Eng, 33(1): 21-30.
- Haefeli, UO et al. (2009), Mol Pharm, 6(5): 1417-1428.
- Mahmoudi, M et al. (2009), J Colloid Interface Sci, 336(2): 510-518.
Iron and iron oxides are natural components of the earth's crust. Iron oxides are extracted from iron ore and can then be converted to iron. Both iron and iron oxides are used in numerous applications both in nanoscale and in larger form. Therefore, there are different ways of release into the environment.

Print toner powder contain iron nanoparticles which can be released unintentionally © Diegofamu/ Fotolia.com
In the environment, it is difficult to distinguish between the natural occurrence of iron and iron oxide nanoparticles and the released nanomaterials generated by human activities. The release of the nanoparticles into air, water and soil is possible. In addition to iron oxides from the earth's crust, insoluble iron oxides also occur in natural waters, e.g. when released by ore mining activities. For example, coalmine wastewater contains many naturally occurring iron nanoparticles [1].
Iron and iron oxide nanoparticles are used in environmental technology for the remediation of groundwater pollutants After the degradation of the harmful chemicals, the nanoparticles remain in the aquifer. Due to the nanoparticles low mobility, it is assumed that they remain at the place of use (see cross cutting text – nanomaterials for environmental remediation) [2,3].
Due to their magnetic properties, iron oxide nanoparticles (such as magnetite) are suitable as carrier materials for catalytically active coatings in wastewater treatment. Since the iron oxide nanoparticles are firmly incorporated into the coating or can be magnetically separated from the water after the treatment, it is unlikely that they will be released into the environment[4].
Unintentional release of iron nanoparticles e.g. into the air via diesel exhaust gases or into the environment from medical applications via wastewater may occur. Iron and iron oxide nanoparticles can also be released into the air from toner dust or printing ink. This takes place mostly in closed buildings, from which, however, a part can escape into the ambient air [5].
Currently it is not possible in the environment to distinguish between technically produced iron and iron oxide nanoparticles and the materials’ natural background thereby hindering the measuring of a corresponding environmental concentration. Consequently, such environmental concentrations were calculated using different computer models including the different release scenarios (see cross cutting topics - Estimating the occurrence of nanomaterials in the environment). These estimated environmental concentrations of iron and iron oxide nanoparticles are much lower than the concentrations that can cause harmful effects in environmental organisms [6].
Engineered iron and iron oxide nanoparticles are released into the environment from various applications. They are difficult to distinguish from the naturally occurring particles of the material. Using computer models, iron and iron oxide nanoparticles pose only a low risk to the environment according to the current state of knowledge.
Literature
- Matthies, R et al.(2012), J Environ Monit, 14:1174-1181.
- Phenrat, T et al. (2009), Environ Sci Technol, 43(1): 195-200.
- Phenrat, T et al. (2007), Environ Sci Technol, 41(1): 284-290.
- Hildebrand, H et al. (2009), Environ Sci Technol, 43(9): 3254-3259.
- Liati, A et al. (2013), Environ Sci Technol, 47:14495-14501.
- Wang, Y et al. (2016), Nanotoxicology, 10(10):1743-5404.
Iron oxide nanoparticles (so-called SPION) are mainly used in medicine, which means that the importance of the main entry ports lungs, skin and gastrointestinal tract for nanoparticles into the body is shifting into the background.
Due to their superparamagnetic properties, iron oxide nanoparticles are mainly used as contrast agents in magnetic resonance tomography (MRT) . Several variants of dextran-coated SPION have been approved already.
Moreover, these special properties can offer the advantage of selective SPION distribution in the body. The SPION can be fixed in the desired target region (the tumor, for example) by means of an external magnetic field. Once fixed, the particles can release attached drugs or substances to the respective area, thus reducing the dose of active agents and minimizing the systemic effects (side effects in the remaining organism) [2,3,4,5,6]. Besides, SPION can be heated through oscillating magnetic fields. This method is used already in cancer therapy. Without damage to the surrounding tissues, tumor cells are killed selectively through local heat development of SPION placed in the tumor [1,7,8].
Literature
- Neuberger, T et al. (2005), J Magn Magn Mater, 293(1): 483-496.
- Torchilin, VP (2000), Eur J Pharm Sci, 11 Suppl 2(0): S81-91.
- Luebbe, AS et al. (1996), Cancer Res, 56(20): 4686-4693.
- Luebbe, AS et al. (2001), J Surg Res, 95(2): 200-206.
- Stroem, V et al. (2004), Nanotechnology, 15(5): 457-466.
- Rudge, S et al. (2001), J Control Release, 74(1-3): 335-340.
- Jordan, A et al. (1997) in U. Haefeli, et al. (Eds.), Scientific and Clinical applications of Magnetic Carriers, Plenum Press, New York, 1997, p. 569-595.
- Jordan, A et al. (1999), J Magn Magn Mater, 201(1–3): 413-419.
- Das, M et al. (2009), Small, 5(24): 2883-2893.
- Hamm, B et al. (1994), J Magn Reson Imaging, 4(5): 659-668.
- Reimer, P et al. (2003), Eur Radiol, 13(6): 1266-1276.
Iron as a vital trace element ensures that animals and plants grow well and stay healthy. However, excessive levels of iron can also have harmful effects on organisms. Iron reacts easily with oxygen ("rusting"), forming iron oxides. Both iron and iron oxide nanoparticles can have different effects on environmental organisms due to their different properties.
 In low concentrations, iron and iron oxide nanoparticles cause no negative effects in bacteria and even promote growth. However, high concentrations trigger negative effects and inhibit growth. One reason for the toxicity of iron or iron oxide nanoparticles is the emergence of ROS [1-5].
In low concentrations, iron and iron oxide nanoparticles cause no negative effects in bacteria and even promote growth. However, high concentrations trigger negative effects and inhibit growth. One reason for the toxicity of iron or iron oxide nanoparticles is the emergence of ROS [1-5].
 Iron oxide nanoparticles accumulate on the body surface and in the intestine of water fleas and brine shrimps. Detrimental effects of high concentration of nanoparticles are attributed to released ions and oxidative stress [6-7].
Iron oxide nanoparticles accumulate on the body surface and in the intestine of water fleas and brine shrimps. Detrimental effects of high concentration of nanoparticles are attributed to released ions and oxidative stress [6-7].
 High concentrations of iron oxide nanoparticles cause oxidative stress and growth disorders in filamentous and compost worms. This is manifested by growth and respiratory inhibitions as well as skin injuries [8,9].
High concentrations of iron oxide nanoparticles cause oxidative stress and growth disorders in filamentous and compost worms. This is manifested by growth and respiratory inhibitions as well as skin injuries [8,9].

For mussels, the presence of either iron or iron oxide nanoparticles in the water is irrelevant. The mussel gill cells take in the iron oxide nanoparticles without impairing respiration [10].
 In various fish species, iron and iron oxide nanoparticles also induce oxidative stress in high concentrations and cause stronger effects compared to iron salts, e.g. an increased mortality in zebrafish. The gill cells of rainbow trout and tilapia are not negatively affected by iron oxide nanoparticles [11-15].
In various fish species, iron and iron oxide nanoparticles also induce oxidative stress in high concentrations and cause stronger effects compared to iron salts, e.g. an increased mortality in zebrafish. The gill cells of rainbow trout and tilapia are not negatively affected by iron oxide nanoparticles [11-15].
 In fruit fly larvae, environmentally relevant (very low) concentrations of iron oxide nanoparticles cause no harmful effects. [16]
In fruit fly larvae, environmentally relevant (very low) concentrations of iron oxide nanoparticles cause no harmful effects. [16]
 Iron oxide nanoparticles attach to the surface of green algae. The associated shadowing of the light leads to a reduced growth of the algae, since they depend on the sunlight [17-18]
Iron oxide nanoparticles attach to the surface of green algae. The associated shadowing of the light leads to a reduced growth of the algae, since they depend on the sunlight [17-18]

Iron and iron oxide nanoparticles are used for the remediation of polluted soil and ground water and can therefore be absorbed by plants. Many plants take up these nanoparticles via their roots from water, but not from soil. The incorporated iron and iron oxide nanoparticles can be detected in many parts of the plants (roots, shoots, leaves). Depending on the concentration, iron and iron nanoparticles can influence the plant growth: low concentrations act as fertiliser and increase the plant growth, while high concentrations of nanoparticles inhibit the growth. The harmful effects of high concentrations are explained by the formation of reactive oxygen species [19-29].
In summary, because of their higher reactivity, iron nanoparticles trigger stronger effects in environmental organisms and plants than iron oxides. However, the concentrations at which effects are observed in environmental organisms are high and not comparable to those actually expected in the environment. Thus, the risk for environmental organisms by iron and iron oxide nanoparticles is considered to be low.
Literature
- Lee, C et al. (2008), Environ Sci Technol, 42(13): 4927-4933.
- Marsalek, B et al. (2012), Environ Sci Technol, 46:2316-2323.
- Nguyen, TK et al. (2015), Scientific Reports, 5:18385.
- Otero-Gonzalez, L et al. (2013), Chemosphere, 93:1201-1206.
- Kumar, N et al. (2014), Water Res, 51:64-72.
- Baumann, J et al. (2014), Sci Total Environ, 484:176-184.
- Kwon, D et al. (2014), Coloid Surf B, 122:384-389.
- Gambardella, C et al. (2014), Environ Monit Assess, 186:4249-4259.
- Yang, YF et al. (2016),Chemosphere, 150:615-623.
- Liang, J et al. (2017), Environ Sci Pollut Res, 24:2507-2514.
- Kadar, E et al. (2010), Anal Bioanal Chem, 396(2): 657-666.
- Hildebrand, H et al. (2010), Environ Pollut, 158(1): 65-73.
- Chen, J et al. (2011), Water Res, 45:1995-2001.
- Zhu, X et al. (2012), PLoS ONE, 7(9):e46286.
- Ates, M et al. (2016), Aquatic Toxicol, 177:22-32.
- Vega-Alvarez, S et al. (2014), Int J Nanomed, 9:2031-2041.
- Toh, PY et al. (2016), Int J Phytoremediation, 18(6):643-650.
- Jagadeesh, E et al. (2015), Colloid Surf B125:284-290.
- Ghafariyan, MH et al. (2013), Environ Sci Technol, 47:10645-10652.
- Trujillo-Reyes, J et al. (2014), J Hazard Mater, 267:255-263.
- Canivet, L et al. (2015), Ecotoxicol Environ Safe, 113:499-505.
- Libralato, G et al. (2016), Ecotoxicol Environ Safe, 1123:81-88.
- Kim, JH et al. (2014), Environ Sci Technol, 48:3477-3485.
- Barhoumi, L et al. (2015), Environ Contam Toxicol, 68:510-520.
- Hu, J et al. (2017), Environ Pollut, 221:199-208.
- Li, J et al. (2016), Chemosphere, 159:326-334.
- Yang, Z et al. (2015), Int J Environ Res Public Health 12:15100-15109.
- Gui, X et al. (2015), Environ Sci Pollut Res, 22: 17716-17723.
- Wang, J et al. (2016), Environ Pollut, 210:338-345.
- Further evaluated literature for the nanomaterial iron & iron oxide can be found here (PDF, 76 KB)
Direct administration into the body (e.g. by intravenous injection into the bloodstream) leads to a rapid and intentional distribution of the particles throughout the body.
Intravenous Administration and Distribution in the Body
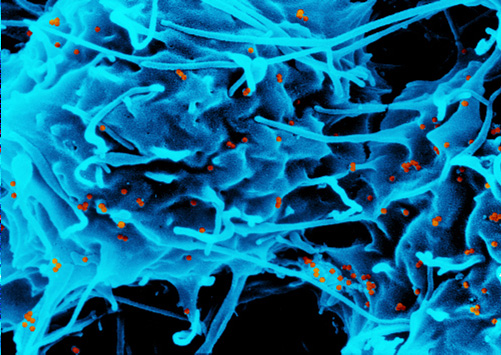
Macrophage filled with iron particles (red). © KIT
Intravenously injected SPION are mainly taken up by macrophages. In these cells, the polymer coating is degraded and is then excreted via the urine or the stool. The iron contained in the SPION is built into the body’s iron stores. The low toxicity of SPION is not surprising considering that natural iron concentrations in the body may be as high as approximately 4000 mg/adult person while no more than 50 – 200 mg/person are administered during medical treatment.
SPION that are provided with a special polymer coating (polyethylene glycol – PEG) are taken up less readily by the macrophages. The retention time of SPION in the body is prolonged through such modification, and the probability of reaching the desired target (for example, the tumor) is increased. Additional protein coatings direct the particles to different organs or cells (for example, folate-coated SPION can be used for marking cancer cells).
The superparamagnetic properties of SPION offer further advantages regarding the selective distribution of the particles in the body. The SPION can be fixed in the desired target region (the tumor, for example) by means of an external magnetic field. Once fixed, the particles can release attached drugs or substances to the respective area, thus reducing the dose of active agents and minimizing the systemic effects (side effects in the remaining organism) [2,3,4,5,6]. Besides, SPION can be heated through oscillating magnetic fields. This method is used already in cancer therapy. Without damage to the surrounding tissues, tumor cells are killed selectively through local heat development of SPION placed in the tumor [1,7,8].
Literature
- Neuberger, T et al. (2005), J Magn Magn Mater, 293(1): 483-496.
- Torchilin, VP (2000), Eur J Pharm Sci, 11 Suppl 2(0): S81-91.
- Luebbe, AS et al. (1996), Cancer Res, 56(20): 4686-4693.
- Luebbe, AS et al. (2001), J Surg Res, 95(2): 200-206.
- Stroem, V et al. (2004), Nanotechnology, 15(5): 457-466.
- Rudge, S et al. (2001), J Control Release, 74(1-3): 335-340.
- Jordan, A et al. (1997) in U. Haefeli, et al. (1997), Scientific and Clinical applications of Magnetic Carriers, Springer Verlag, p. 569, ISBN
9780306456879. - Jordan, A et al. (1999), J Magn Magn Mater, 201(1–3): 413-419.
- Mueller, K et al. (2007), Biomaterials, 28(9): 1629-1642.
Behaviour at the Blood-Brain Barrier
Superparamagnetic particles with special coatings, the so-called "solid-lipid nanoparticles" (SLN), were injected in the bloodstream of rats. It was shown that these modified SPION pass the blood-brain barrier, accumulate in the brain, and remain there over a sufficiently long period of time to record MRT data of the brain [1]. In tumor therapy, the barrier is avoided by injecting the iron oxide particle solution directly into the tumor tissue of the brain.
Literature
- Peira, E et al. (2003), J Drug Target, 11(1): 19-24.
Behaviour of Uptake in somatic cells
After intravenous administration, the SPION particles are mainly taken up in the liver by special macrophages (the so-called mononuclear phagocytosis system). In these, larger SPION (approximately 100nm) are taken up via receptor-mediated endocytosis while smaller ones (< 20nm) get into the cells through pinocytosis. In addition, it was shown that plasma proteins in the blood bind to SPION particles, thus influencing uptake of the latter in the cells. To better understand the uptake mechanism in the cell and the distribution of the particles in the body, it is important to identify which proteins bind to the nanoparticles.
Literature
- Schulze, E et al. (1995), Invest Radiol, 30(10): 604-610.
- Raynal, I et al. (2004), Invest Radiol, 39(1): 56-63.
- Fleige, G et al. (2002), Invest Radiol, 37(9): 482-488.
- Roecker, C et al. (2009), Nat Nanotechnol, 4(9): 577-580.
- Ehrenberg, MS et al. (2009), Biomaterials, 30(4): 603-610.
Iron nanoparticles oxidise very quickly under environmental conditions, and are consequently transformed into iron oxide nanoparticles. A coating can prevent the oxidation process of the nanoparticles. Uncoated, synthetically produced iron oxide nanoparticles behave similarly to naturally occurring iron nanoparticles.

oxidized tailpipe © UFZ
In general, the mobility of nanoparticles in soil or water depends on whether they are isolated, form agglomerates or react with other compounds present in the environment .
In soil, the transport of iron or iron oxide nanoparticles is depending on particle size. Even single nanoparticles are not widely transported in soil. Investigations of soil and water remediation activities with iron nanoparticles have shown that the introduced nanoparticles move less than one meter away from the injection site [1] .
For the behaviour of nanoparticles in aqueous environments, the waters’ composition is important. The pH value, salinity and the content of organic material influence the agglomeration and sedimentation process of iron and iron oxide nanoparticles. The presence of high concentrations of organic material in the water can prevent the agglomeration of the nanoparticles. However, high concentrations of calcium in the water favour the agglomeration of the nanoparticles resulting in the sedimentation of the iron and iron oxide nanoparticles [2-5,12,13].
Ground water remediation processes make use of iron nanoparticles with different coatings, which prevent nanoparticle agglomeration thus keeping the iron nanoparticles more reactive and mobile [1,11,12].
Naturally occurring iron oxide nanoparticles are able to bind and transport harmful compounds like copper complexes. This behaviour can also be exploited for synthetically produced iron oxide nanoparticles. In heavily polluted waters, iron oxide nanoparticles bind e.g. arsenic and mercury, thereby reducing the heavy metal content in the water and thus reducing the risk for environmental organisms.
Other pollutants present in the water, such as aromatic hydrocarbons (e.g. phenanthrene and naphthalene), can also be captured by the iron and iron oxide nanoparticles and removed from the water. Furthermore, iron and iron oxide nanoparticles facilitate the degradation of pollutants like benzo(a)pyrene in soil and water, which is purposely used in remediation [6-8,14-17].
The surface of iron and iron oxide nanoparticles oxidises quickly leading to the formation of iron oxide nanoparticles. This oxidation process occurs in all environmental compartments. Engineered iron or iron oxide nanoparticles behave almost identical to naturally occurring nano- and micro scale iron and iron oxide nanoparticles. Their properties depend on the conditions of the surrounding environment such as pH value or salinity and can be influenced by the binding of organic compounds. Furthermore iron oxide nanoparticles can capture and remove pollutants from the environment.
Literature
- Phenrat, T et al. (2009), Environ Sci Technol, 43(13): 5079-5085.
- Baalousha, M et al. (2008), Environ Toxicol Chem, 27(9): 1875-1882.
- Baalousha, M (2009), Sci Total Environ, 407(6): 2093-2101.
- Zhang, Y et al. (2008), Water Res, 42(8-9): 2204-2212.
- Zhang, Y et al. (2009), Water Res, 43(17): 4249-4257.
- Hochella, MF et al. (2005), Geochim Cosmochim Ac, 69(7): 1651-1663.
- Sherman, DM et al. (2003), Geochim Cosmochim Ac, 67(22): 4223-4230.
- Fang, J et al. (2008), Langmuir, 24(19): 10929-10935.
- Buchkowski, RW et al.(2016), Bull Environ Conam Toxicol, 96:83-89.
- Liu, A et al. (2015), Chemosphere, 119:1068-1074.
- Greenlee, LF et al. (2012), Environ Sci Technol, 46:12913-12920.
- Dong, H & Lo, IMC (2013), Water Research, 47:419-427.
- Chekli, L et al. (2013), Sci Total Environ, 461-462:19-27.
- Hartland, a et al. (2015), Environ Sci Technol, 48:14101-14109.
- Gil-Diaz, M et al. (2017), Sci Total Environ,584-585:1324-1332.
- Gupta, H & Gupta, B (2015), Chemosphere, 138:924-931.
- Kim, E-J et al. (2014), Sci Total Environ 470-471:1553-1557.
 >
>