 >
Spotlight February 2021: Nanoobjects in the COVID-vaccine – scientifically correct?
>
Spotlight February 2021: Nanoobjects in the COVID-vaccine – scientifically correct?
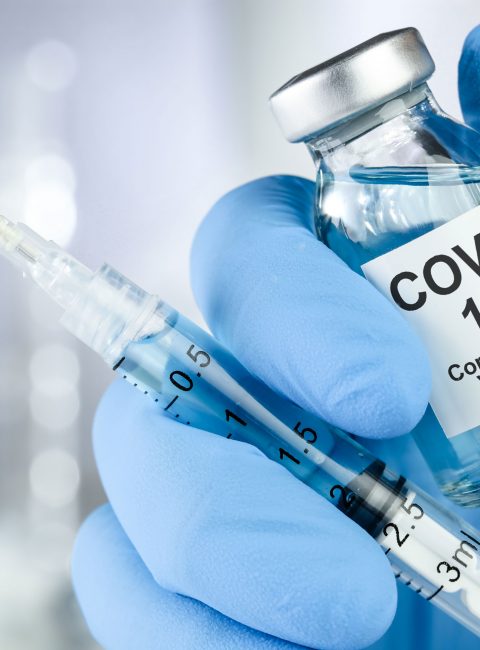
The COVID-19 pandemic induces very different reactions of people on the internet (https://www.cdc.gov/mmwr/volumes/70/wr/mm7002e1.htm) and in the social networks. Without following the conspiracy theories as “5G nanochip hidden in COVID vaccines” some news as “COVID vaccines induce allergic reactions” should be scientifically recognised. The picture from the 5G-nanochip whose plan goes viral on the internet is now exposed as fake and shows a guitar pedal. Quite different the recent news about allergic responses after injection of the COVID vaccine [1-3]. Is it true, and how about nanoparticles in vaccines?
Firstly, we should know that the active ingredient within the new vaccines are very sensitive molecules (the so-called mRNA) which must not only be cooled during transport but also would immediately be degraded after injection if not explicitly be stabilized by a special nano-shell. Here the principles of nanomedicine are adopted as the instable RNA-molecule is packed into a lipid layer containing stabilizing polymers (e.g. polyethylene glycol = PEG and others) [4, 5]. These polymers are part of the transport system which delivers the active agent to the cells in our body which start the anti-viral machinery. Unfortunately, such molecules may have a disadvantage because a small part of the population responds sensitive to polymers such as PEG which has been observed in 12 patients after getting the vaccine [1, 3]. The portion of sensitive people is very small but not negligible (currently more than 32 Mio. people have been vasccinated, as of January 2021), thus, it is important to know about the ingredients [please find the full list of the two recent vaccines in 3] and to discuss these with the treating physician. Scientific fact is: the size of the lipid vesicles (nano) is not of importance in relation to adverse effects. Ingredients such as the polymers of the shell around the active agent may induce problems, but the advantage of a COVID vaccination is indisputable.
If someone wants to know more about nanoparticles and vaccination, please visit the website of the Helmholtz Centre for Infection Research: https://www.helmholtz-hzi.de/en/news-events/stories/vaccination-with-nanoparticles/ .
Literature:
- de Vrieze, J (2021) Pfizer’s vaccine raises allergy concerns. Science, 371(6524): 10-11.
- Kleine-Tebbe, J et al. (2021) Severe allergic reactions to the COVID-19 vaccine – statement and practical consequences. Allergol Select, 5 26-28.
- Worm, M et al. (2021) Covid-19 vaccination and risk of anaphylaxis – Recommendations for practical management. MMW Fortschr Med, 163(1): 48-51.
- Editorial (2020) Nanomedicine and the COVID-19 vaccines. Nat Nanotechnol, 15(12): 963
- Abd Ellah, NH et al. (2020) Nanomedicine as a promising approach for diagnosis, treatment and prophylaxis against COVID-19. Nanomedicine (Lond), 15(21): 2085-2102.
Weitere Spotlights
Spotlight August 2023: From principles to reality. FAIR implementation in the nanosafety community
In the August 2023 Spotlight, we present a paper that addresses the implementation of FAIR (Findability, Accessibility, Interoperability and Reusability) Data in nanosafety research. The authors introduce the new AdvancedNano GO FAIR Implementation Network (see also https://www.go-fair.org/implementation-networks/overview/advancednano/) established as part of the GO FAIR initiative. The paper highlights the AdvancedNano GO FAIR Implementation Network’s support […]
Read moreSpotlight October 2020: Nanosafety – Topic of the Future
Research on nanosafety is a driver of innovation as the spotlight in July has demonstrated. But furthermore, this research field is built on routine as well if researchers look for the “needle in the haystack”. In many areas the safety research initiates the development of new methods, e.g. for the determination of nanoparticles within exposed organisms via […]
Read moreSpotlight April 2022: A new risk assessment of nanomaterials in 3D printing is needed
The use of nanomaterials in 3D printing has great potential. Due to the properties of nanoscale materials, many requirements can be implemented in 3D printing. However, these unique properties based on the size of the particles also lead to the need for new risk assessments. This is because if the nanoparticles are released in the […]
Read moreSpotlight October 2021: Nanopesticides – a proposal for a risk assessment framework
The application of so-called “nanopesticides” (see also cross-sectional text Nanomaterials in plant protection products) is said to have two basic advantages: a smaller amount of pesticide is needed for the same agricultural area and the efficacy is improved. This is necessary to grow enough food for a still growing world population. However, this could also […]
Read more


