The term perovskite summarizes a group of materials that derives from a mineral, the perovskite. The various perovskites have a similar structure, that is, atoms and molecules are arranged similarly. However, they are composed of different atoms or molecules.
This article will mainly discuss perovskites for solar cells, although there are many others.
How can I come into contact with this material
 Because solar cell perovskites are being researched in laboratories ( Status 2022), individuals do not come into contact with these materials at this time. Before solar cells perovskites are launched on the market, it must be ensured that they will not release toxic substances to humans and the environment. However, the perovskites currently being developed release the toxic heavy metals tin and lead, respectively, when they come into contact with water.
Because solar cell perovskites are being researched in laboratories ( Status 2022), individuals do not come into contact with these materials at this time. Before solar cells perovskites are launched on the market, it must be ensured that they will not release toxic substances to humans and the environment. However, the perovskites currently being developed release the toxic heavy metals tin and lead, respectively, when they come into contact with water.
One possible way to prevent the release of lead and tin from solar cell perovskites would be to safely encapsulate them. This could be done by encasing them in glass. However, there are general safety concerns here, because if the glass is damaged, a release of the heavy metals would again be possible. The problem could be solved if the heavy metals in the solar cell perovskites were replaced by harmless elements or compounds, or if the release of toxic components were impossible.
Is there any risk from this material to humans and the environment?
From solar cells perovskites containing tin or lead, the heavy metals tin or lead could be dissolved out. Both heavy metals are toxic to humans and environmental organisms and in some cases are also accumulated in plants, animals and fungi. Therefore, a release of the heavy metals must be prevented. However, a hazard currently exists only theoretically, as these solar cells are not sold for installation on rooftops, so they are not on the market (as of 2022).
If a production or sale of these solar cells containing tin or lead is realized in the future, it must be ensured that no heavy metals are released into the environment or to humans.
Conclusion
Solar cells perovskites are still research materials in 2022. Studies on the toxicity of such materials are useful. There is no danger for private persons at this time, because one cannot buy perovskite solar cells, so one does not come into contact with this material.
By the way
- Perovskites could be the most abundant minerals on Earth, because it is assumed that a whole layer of the Earth’s crust (mantle) is made of perovskite.
- The occurrence of perovskites is not limited to the Earth: they have also been found in meteorites
Properties and applications
Perovskite is a mineral chemically referred to as calcium titanate. It has the chemical formula CaTiO3. Because of its characteristic crystal structure, the mineral perovskite was also used as the name for an entire class of materials, the perovskites, because there are many compounds with the general formula ABC3 that form comparable crystal lattices to the mineral perovskite. Strictly speaking, the mineral perovskite, i.e. calcium titanate, does not crystallise in a perovskite structure at all, but only in a distorted perovskite structure. Perovskites found in nature are mostly composed of divalent and tetravalent ions (e.g. in calcium titanate, CaTiO3 the composition is A = Ca2+; B = Ti4+, C = O2-, Ca = calcium, Ti = titanium, O = oxygen). One can compare the structure with a long-term parking garage with permanently allocated parking spaces, in which the arrangement of the parking spaces is fixed. Which vehicles (= atoms, molecules or ions) are then parked in the car park is initially irrelevant, so in the original perovskite mentioned above, Ca, Ti and O are "parked". Crystallographically, perovskites consist of octahedra linked on all sides, cf. right image. The left-hand image shows that the B ion (yellow) is located in the centre of the octahedron, which is surrounded by C ions (blue) (shown in the front of the left-hand image). Between the octahedra there are the A ions (green), which shift the linked octahedra depending on the size of A and thus disturb the crystal lattice.
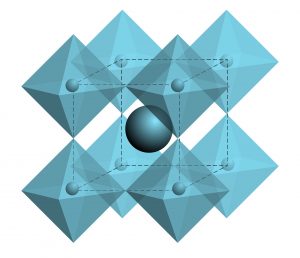
Linked octahedra in perovskite © julia_faranchuk-adobestock
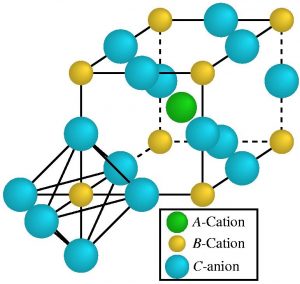
Position of the A, B, and C atoms or molecules in the perovskite - Image source: Lonxos@de.wikipedia.org
This article focuses on the perovskites used in new, innovative solar cells. Such perovskites for solar cells, deviating from the composition described above, are often synthesised from mono- and dications as well as monoanions in the laboratory e.g. (CH3NH3)[PbBr3] i.e. A= CH3NH3+, B= Pb2+, C=Br-, (or if one writes out the molecular formulas: methylammonium tribromoplumbate from A = methylammonium ions, B = lead(II) ions and C = bromide ions).
Since perovskites are a large class of materials, there are a variety of possible applications.
The perovskite materials for solar cells are currently research materials, i.e. these materials developed in research projects are not yet on the market.
Perovskites in solar cells and energy-converting devices
At the beginning of 2022, solar cells made of perovskite materials were the main topic of discussion in the press, because the industry hopes to achieve inexpensive and easy-to-manufacture photovoltaic units here and to be able to dispense with larger quantities of expensive silicon. For the three components A, B and C in the typical ABC3 perovskite, the following ions or compounds are usually mentioned in the literature :
- A: Cs+, CH3NH3+or HC(NH2)2)+ (caesium, methylammonium or formamidinium cations)
- B: Pb2+ or Sn2+ (lead(II)- or tin(II) Ions)
- C: Cl-, Br-or I- (chloride, bromide or iodide ions)
However, materials containing lead may release this toxic element into the environment, which should be prevented urgently . Therefore, the focus of some research has been on tin containing materials . However, these can release tin ions, which can also be toxic.

Perovskite solar cells convert a high proportion of incoming light directly into usable electricity. ( Picture:Fabian Ruf/Scilight)
In addition to the low price of perovskite solar cells, their property of being semi-transparent in a thin layer is particularly interesting. This means that another solar cell can be placed under a perovskite solar cell, which can also convert the remaining transmitted light into electricity. Such solar cells are called tandem solar cells because two energy-converting layers are present.
In addition, in the field of energy conversion, thermoelectrics are also powered by perovskites, which means heat is converted into electricity. If you combine conventional solar cells with thermoelectrics, you get tandem solar modules with two energy-converting layers again. The two individual components, solar cell/photovoltaics and thermoelectrics, then contribute to the efficiency of the solar cells, hence the electricity yield.
If you coat a display with a transparent perovskite solar cell, you could create a power-supplying display.
Other applications of perovskites
Much less visible is the use of perovskites such as barium titanate (BaTiO3, Ba = barium, Ti = titanium, O = oxygen) as dielectrics in capacitors or as ferroelectrics. The charge separation capability in perovskites is also used for these applications.
In the field of energy transport, the YBCO (chemical formula YBa2Cu3O7-x, Y = yttrium, Ba = barium, Cu = copper, O = oxygen) superconductor, which crystallises in a distorted perovskite structure, has achieved a certain prominence. The abbreviation YBCO is a simplified description of the most popular and relatively easy to produce so-called high-temperature superconductor (production only under the supervision of experts!), which already becomes superconducting when cooled with liquid nitrogen (-196°C).. The YBCO was a further development of the superconductor with the formula La2-xBaxCuO4, (La = lanthanum) which was awarded a Nobel Prize in Physics in 1987 and describes the first representative of the class of ceramic high-temperature superconductors.
These applications show that perovskites can be innovative materials. Nanoscale perovskites are also used in the field of thermoelectrics.
Furthermore, perovskites are discussed as coating materials for implants, e.g. in dental prostheses, to improve their biocompatibility . Other applications of perovskites are described in the literature for wastewater treatment, adsorbents or catalysts, and bioimaging .
Origin and production

Perovskite mineral © vvoe - stock.adobe.com
Perovskites occur naturally in various rocks. It is remarkable, for example, that a large part of the Earth's mantle could consist of a silicate perovskite, which would make it the most common mineral on Earth. It is formed under immensely high pressure at a depth of several kilometres in the Earth's mantle .
All perovskites used in solar cells, thermoelectrics, dielectrics or superconductors do not occur naturally. They are exclusively produced in the laboratory. An exception are the perovskites used for coating materials, e.g. CaTiO3 (calcium titanate): although this occurs naturally, laboratory-produced CaTiO3 is used for implant coating. It can thus be shaped directly into the form needed for the dental treatment. Moreover, synthetically produced CaTiO3 is usually much purer than naturally obtained CaTiO3.
Since perovskites are not a single material but a class of materials, there is also no uniform method of production .
Current perovskite materials developed for the application in solar cells belong to the group of so-called "research materials", meaning these materials developed in research projects are not yet on the market. Depending on their composition, perovskite materials can be used in innovative solar cells or in the medical field as bone replacement materials or biosensors. .
 For innovative solar cells, lead or tin based perovskites are currently being investigated as active components. However, the heavy metal lead is a known toxic substance, which is why direct contact with humans, e.g. via accidental release, should be avoided at all costs. Therefore, more environmentally friendly materials such as tin, germanium, magnesium, manganese or nickel are being tested as alternatives for perovskites in solar cells. In addition, encapsulation techniques are being developed to prevent release of the lead or other active components from the perovskites.
For innovative solar cells, lead or tin based perovskites are currently being investigated as active components. However, the heavy metal lead is a known toxic substance, which is why direct contact with humans, e.g. via accidental release, should be avoided at all costs. Therefore, more environmentally friendly materials such as tin, germanium, magnesium, manganese or nickel are being tested as alternatives for perovskites in solar cells. In addition, encapsulation techniques are being developed to prevent release of the lead or other active components from the perovskites.
Everyday Contact
In everyday life, the consumer does not yet come into contact with the perovskite materials for solar cells, since no products with these materials are yet on the market (as of 2022). In addition, the perovskites are firmly embedded in the solar cells or processed accordingly, so that no release takes place under normal conditions. However, possible damage to the solar cells due to aging or external influences such as hail, heavy rain or fire could promote the release of the perovskite materials or their degradation products, including lead ions, from the product.
Situation at the Workplace
The biggest chance of contact with perovskite materials designed for solar cells or their individual components exists for researchers at the workplace. This applies to the entire life cycle of the solar cells, from the manufacture of the lead or tin containing perovskites, to the production and ultimate disposal of the solar cells. For the handling of toxic substances such as the heavy metals lead or tin at the workplace, common safety regulations (e.g. for lead BAuA TRGS 505 - Handling of Lead) and occupational exposure limits (European Chemicals Agency - ECHA) apply. In the EU, an air limit value at the workplace of 0.15 mg/m3 applies when handling lead and inorganic lead compounds (98/24/EC, DGUV).
To estimate the potential risk of lead containing perovskites, laboratory studies were carried out with cell lines representing the lung (cell lines A549, BEAS-2B), liver (cell line HepG2) or the digestive tract (cell line Caco-2). Under the experimental conditions (24 h), up to 30% of the initial lead was dissolved as ions from the perovskite material, depending on the material type. Minor damage or inhibition of cell growth could be observed, although the cell lines investigated reacted with varying sensitivity in the experiments. However, the observed cell damage is several times lower than that observed when exposed to pure lead ions. Presumably, the observed effects of the perovskites are due to the released lead ions, but this needs to be further investigated.
Consumer Situation
Currently (as of 2022), perovskites are still research materials and not yet used in commercially available solar cells. Lead or tin containing perovskites are firmly embedded in these solar cells so that there is no contact with humans during normal handling. However, if such solar cells are damaged, e.g. by aging or external influences such as hail, heavy rain or fire, the bound perovskites or their degradation products, including lead or tin ions, may be released into the environment. The EU Directive 2011/65/EU, also known as RoHS directive (Restriction of hazardous substances), regulates and restricts the use of certain hazardous substances in electrical and electronic equipment. The aim is to minimize the input of toxic substances such as lead, chromium or cadmium from electronic waste.
During normal handling of perovskite solar cells, humans do not come into contact with the perovskites containing lead or tin. However, due to damage or aging of the solar cells, the embedded materials or their degradation products, which also include lead or tin ions, can be released into the environment and humans can indirectly come into contact with them. Several strategies are currently being pursued to reduce or prevent such releases by encapsulating the toxic ingredients or replacing them with more environmentally friendly alternatives. However, solar cells with perovskite materials are still in the research stage and are not available as products on the market.
Further Information
- European Chemicals Agency (ECHA). Occupational exposure limits substance evaluations “Lead and its compounds" (last accessed: 09/2022)
- Federal Institute for Occupational Safety and Health (BAuA) (2021). TRGS 505 Lead - Technical Rule for Hazardous Substances, Edition: March 2021, GMBl 2021 pp. 582-598 [No. 26] (of 4 May 2021), Revised: GMBl 2022 p. 512 [No. 22] (of 1 July 2022) (unofficial version; mandatory is the current German version)
- Institute for Occupational Safety and Health of the German Social Accident Insurance (IFA DGUV). Binding occupational exposure limit values (OELs) of the European Commission (last access: 09/2022)
- European Commission (2911). Directive 2011/65/EU of the European Parliament and of the Council of 8 June 2011 on the restriction of the use of certain hazardous substances in electrical and electronic equipment (recast) Text with EEA relevance, OJ L 174, 1.7.2011, p. 88–110
Innovative perovskite solar cells contain lead or tin compounds as active components. However, due to weathering or environmental damage from hail, storms, fire or heavy rain, the heavy metals, along with various acids, could be released from the solar cells and enter the environment. Therefore, such perovskite solar cells are sealed to minimize or prevent the release of heavy metals. The perovskite materials for solar cells are currently so-called "research materials", i.e. these materials developed in research projects are not yet on the market.
Release of perovskite

Danger from thunderstorms, heavy rain, storms © trendobjects-stock.adobe.com
During the life cycle of solar cells, the heavy metals contained could escape during the production of the materials or the finished product, use or disposal. The release during production can be reduced by safety measures. During use and disposal, on the other hand, environmental influences play a major role. It is therefore generally important to seal the solar cells or encapsulate the active components in order to prevent heavy metals and other active components from escaping as a result of fire, explosion or at the end of their service life if they are carelessly dumped. Even slight damage should be prevented. Otherwise the perovskites come into contact with the humid ambient air and oxygen. This can lead to degradation or oxidation of the perovskites. As a result, the heavy metals lead and tin can be released into the air (in the event of a fire) or into the water and get into the ground via rainwater runoff. Acids formed during degradation, such as hydroiodic acid, also cause a change in the pH value in water and soil. Released heavy metals can subsequently accumulate in soil and water.
The escape of heavy metals from solar cells at the end of their useful life can be prevented by correct disposal or further recycling can be controlled in a targeted manner. With appropriate recycling, the heavy metals can be recovered and reused. Whether solar cells are declared as toxic or non-toxic waste depends on the heavy metal content and the likelihood of release, as well as on national waste disposal regulations. Should perovskite materials for solar cells come onto the market, the corresponding regulations must be taken into account.
Released amount of perovskite
Perovskite solar cells containing lead or tin are currently purely research materials and are not yet available on the market. Environmental hazards from the release of heavy metals and acids from such perovskite solar cells were therefore only investigated under laboratory conditions. After simulating events such as heavy rain, flooding or fire, the release of the heavy metals lead and tin was measured. Perovskite solar cells containing lead or tin can be damaged by environmental influences and the heavy metals they contain can be released. The penetrating oxygen and water result in bromine or iodine-containing carbon compounds (organo-halide compounds) iodine, which further decompose into acids and nitrogen compounds (amines). Heavy metal halide compounds (i.e. lead or tin compounds containing bromine or iodine) are also formed. Furthermore, dissolved lead-containing perovskite nanoparticles can release lead in the form of ions.
The lead content of perovskite solar cells is known. A lead-containing perovskite solar cell module contains about 0.8-0.9 g of lead. There is no information on the tin content.
The release of lead compounds in the form of lead iodide from perovskite-containing foils for solar cells through rain is estimated to be moderate. Lead is poorly soluble in water. Even acid rain, which would favor faster release, does not lead to a significant increase in lead iodide release (Hailegnaw, Kirmayer 2015). In general, an accumulation of lead in the upper layers of the earth or small, closed groundwater reservoirs cannot be ruled out for heavy rain events.
Perovskite solar cells containing lead can be damaged by flooding. The lead can enter the environment in the form of lead iodide, although the exact amount of lead iodide released is unknown.
Extreme solar radiation and thus increased temperatures of 45°C lead to the release of lead iodide from perovskites. At a lower ambient temperature of 25°C, on the other hand, there is no release. Bromine-containing perovskites, on the other hand, show signs of decomposition neither at 45°C nor at 25°C .
The effect of fire on leaded perovskite solar cells was also studied. The bound lead breaks down into lead iodide during combustion and is released into the air. Less lead was released in the form of lead oxide from perovskite solar cells encapsulated with glass. This type of encapsulation is therefore an effective measure to reduce the release of lead. No statement was made on the amount of lead that escaped. When disposing of old perovskite solar cells containing lead, around 70% of the bound lead is released within a year of being dumped. When old lead-containing perovskite solar cells are incinerated, almost all of the lead ends up in the ash, while other toxic substances are released through the incineration process. The recycling of perovskite solar cells and the recovery of the heavy metals contained is therefore preferable.
In general, the amount of heavy metals released from perovskite solar cells depends heavily on the environmental conditions. For example, the pH value and naturally occurring acids (humic acids) in the soil play a role in the solubility and agglomeration behavior of the materials.
The studies carried out to date on the release of heavy metals and acids from perovskite solar cells do not yet provide a uniform picture, so that the quantities released cannot be estimated. Therefore, no statement can yet be made as to whether existing limit values at national or international level for the lead content are exceeded, as has already been observed for silicon-based solar cells and thin-film panels. However, the release of heavy metals should be as low as possible while maintaining the functionality and efficiency of the solar cells, and work should continue on effective methods for encapsulating potentially critical substances. In the future, the advantage of using renewable energies must also be compared with the emissions that occur when fossil fuels are burned.
Perovskite solar cells can be damaged by various environmental influences and consequently release heavy metal compounds and acids. These can get into the air, water and soil. The amount of release depends on many factors, such as the number or area of damaged solar cells and the type and intensity of the environmental influences. Such innovative perovskite solar cells are currently still under development and all data on the release come from laboratory experiments or simulations.
Further information
The development of innovative solar cells with perovskite materials as active components is still at the research stage and there are no products on the market yet (as of 2022). In the workplace, researchers may inadvertently take up released perovskites for solar cells into the body through inhalation or skin contact. Applicable workplace protection measures as well as air and occupational exposure limits are designed to protect workers from contact with known toxic substances such as lead or tin. In addition, perovskites or their individual components such as lead or tin could be released into the environment from damaged solar cells and absorbed by humans through the food chain via the digestive tract. Initial laboratory studies with lead containing perovskites for solar cells indicate that the observed mild toxic effects are caused by released lead ions.
Uptake via the lung
In the production and processing of lead or tin containing perovskites for solar cells, inhalation of the materials is a critical route of exposure. For the handling of heavy metals such as lead or tin, regulations and occupational limit values are in place to reduce or prevent exposure. Lead as well as its potentially more environmental friendly alternative tin, are known to be toxic even in low concentrations.
In laboratory experiments with lung cells, the possible damage of lead containing perovskites was investigated in comparison with their lead containing degradation products and pure lead ions. In various lung cells, no (cell line A549) or only slight damage (cell line BEAS-2B) was detected after 24 h exposure to lead containing perovskites or their degradation products. Agglomeration of the perovskites took place in the respective culture media. Up to 30% of the initial lead content of the perovskites could be detected as lead ions in the culture medium after 24 h. The observed minor damage is likely due to the released lead ions. The underlying mechanisms need to be further investigated.
Since the investigated lung cell lines showed different sensitivities to the tested perovskite materials, further investigations are necessary to conduct a comprehensive risk assessment.
Uptake via the skin
Skin contact can be prevented by wearing suitable protective clothing when handling lead or tin containing perovskites for solar cells. Explicit studies on skin exposure to perovskites are currently (as of 2022) not available.
Uptake via the gastro-intestinal tract
An uptake of lead or tin containing perovskites for solar cells via the digestive tract is very unlikely or only possible via indirect intake. Perovskites or their degradation products could be released unintentionally from the solar cells into the environment and enter the human digestive tract via the food chain.
To assess possible effects, laboratory studies were performed on intestinal cells (cell line Caco-2) with different perovskite solar cell materials. The intestinal cells showed a slight concentration-dependent toxicity to the perovskite particles. They also react more sensitively to the perovskites, i.e. effects already occur at lower concentrations compared to the studied lung cells. The toxic effect can possibly be attributed to the lead ions released from the materials. However, further investigations are necessary for more detailed conclusions or understanding of underlying mechanisms.
The development of perovskite based solar cells is still at the research stage and there are no products yet on the market (as of 2022). An uptake of such perovskite materials into the body is only very low or relevant for the situation at the workplace. Inhalation of released particles during production or processing can be minimised or prevented by appropriate protective measures. Initial studies in the laboratory indicate that possible toxic effects of lead containing perovskites are due to the release of lead ions. Appropriate approaches to minimise the release of harmful heavy metal ions or replacement with less harmful materials are currently under scientific investigation.
Further Information
- Agency for toxic substances and disease registry (ASTDR) - Toxicological Profile for Lead (Last Accessed: 09/2022)
- Agency for toxic substances and disease registry (ASTDR) - Toxicological Profile for Tin (Last Accessed: 09/2022)
The greatest threat to environmental organisms from perovskite solar cells comes from the heavy metals they contain, such as lead or tin, as well as from various acids. The effects of lead and tin on environmental organisms have been well studied and critical concentrations are known. This data can be used to provide an assessment of the risk to environmental organisms from perovskite solar cells. Currently (as of 2022), perovskites are still purely research materials and are not installed in solar cells or are available on the market as products.
Release of perovskite in environmental organisms
Under normal circumstances, environmental organisms do not come into direct contact with the heavy metals contained in intact perovskite solar cells (on roofs or in solar parks). However, environmental influences such as fire, storms or flooding could damage the solar cells and as a result a number of toxic heavy metals and acids could escape. Environmental organisms can come into contact with and absorb these heavy metals and acids via the air, water or soil.
Toxicity of perovskite in environmental organisms
In particular, various degradation products from perovskite solar cells containing lead or tin (heavy metals, acids and ammonium compounds) can have a toxic effect on environmental organisms. An important mechanism is the formation of hydroiodic acid and the associated reduction in pH (acidification) in an aqueous environment. This acidification is particularly harmful to aquatic organisms. Lead ions released from damaged solar cells or materials can also be toxic to environmental organisms. These degradation processes can also be observed in laboratory studies with water fleas. During degradation in an aqueous environment, the hydroiodic acid formed from lead-containing perovskites lowers the pH value of the water and increased mortality of the water fleas has been observed. However, compared to the toxicity of pure lead, the observed effects are lower. Accumulation of ingested lead in water fleas has been observed.
Perovskite nanoparticles containing lead administered via the water or feed cause concentration-dependent toxicity in zebrafish. This effect does not differ in comparison to pure lead and can therefore be attributed to the lead ions released. This is confirmed by further investigations with lead iodide , and leaching products from mechanically destroyed perovskite solar cells. In contrast to pure lead, lead-containing perovskite nanoparticles ingested through food cause a change in the activity of genes in the gut of zebrafish. The amount of heavy metals released from perovskite solar cells containing lead or tin leads to moderate toxicity in zebrafish embryos. This means that the spilled amounts of lead and tin can damage aquatic organisms, but are not fatal. However, a prerequisite for this assessment is that the released lead is distributed evenly in the environment and that there is no locally increased concentration, for example in the upper soil layer or in a small groundwater reservoir. The investigations also show that tin released from damaged perovskite solar cells is more harmful to fish than the released lead. However, as a pure heavy metal, lead is more toxic than tin. Other released substances, such as acids, may also play a role in the damaging effect. Perovskite solar cells containing tin are therefore not an environmentally friendly alternative to perovskite solar cells containing lead. In comparison, fish are less sensitive to lead-containing perovskites than water fleas.
Lead ions released from perovskites containing lead are quickly absorbed by algae and released back into the environment after longer cultivation. Very small amounts of lead-containing perovskites (up to 5 mg/L) can promote algae growth, while larger amounts (40 mg/L) inhibit it. The reason for this is an inhibition of chlorophyll and protein formation.
Aquatic bacteria are more sensitive to lead-containing perovskites than soil bacteria or other environmental organisms. In both types of bacteria, the lead-containing perovskites reduce metabolism and growth. These effects are mainly attributed to the released lead ions.
The effects on soil bacteria are influenced by transformation processes of the lead-containing perovskites in an aqueous environment. The pH value and the presence of humic acids or other cations such as magnesium or calcium influence the solubility and aggregation behavior of perovskites in an aqueous environment. Increasing the pH or humic acid concentration decreased the toxicity of the perovskite materials by reducing the release of lead ions. In contrast, the presence of magnesium or calcium in combination with humic acid enhanced toxicity due to increased release of lead ions.
Bound heavy metals in the form of heavy metal halides, such as those released from perovskite solar cells, pose a particular threat to plants. Plants absorb the lead compounds released into the soil and the plants accumulate. In the examined plants, peppermint, chili and cabbage, the lead halides from perovskite building blocks (lead iodide and methylammonium iodide) accumulate 10 times more effectively compared to the natural occurrence of lead. An accumulation of lead via the food chain is therefore also possible. Tin and tin compounds from perovskite solar cells accumulated less in plants (Li, Cao et al. 2020). For peppermint plants exposed to tin(II) iodide via the soil, toxicity was lower than for lead, because less hydroiodic acid is formed in the soil.
In addition to the perovskite solar cells containing lead or tin, which are still under development, there are solar cells that have been in use for many years. These classic solar cells are roughly divided into crystalline solar cells (consisting of ordered silicon crystals) and thin-film panels (evaporated silicon). Classic solar cells also contain small amounts of heavy metals, which can have a toxic effect on environmental organisms. In the case of crystalline solar cells, lead and tin are primarily responsible for the toxic effect. In the case of thin-film panels, it is mainly lead and chromium. Algae reacted most sensitively to the heavy metals. In a direct comparison to lead-containing perovskite solar cells, damaged silicon-based solar cells showed higher toxicity to zebrafish and water fleas. These are comparative laboratory studies in which extreme events are simulated. Entire solar cells were crushed, exposing the organisms to extremely high concentrations of heavy metals. These concentrations are not to be expected under environmental conditions.
Assessment of the effects of perovskite in environmental organisms
 Perovskites containing lead and tin do not pose a threat per se to aquatic organisms and plants because the toxic compounds are bound or encapsulated in the product. Substances that can be released unintentionally due to environmental influences during the life cycle of the perovskite solar cells containing lead or tin are to be evaluated as critical. These include above all heavy metals, acids and other substances that have not yet been identified. The level of toxicity of the pollutants depends on the solubility of the substances, but also on the pH of the environment (water, soil). Since these innovative perovskite solar cells are currently still in the research stage, there is only little data from the laboratory on the release quantities. Heavy metals themselves have already been well investigated toxicologically and existing limit values can be used in the future to better assess the risk to organisms from perovskite solar cells.
Perovskites containing lead and tin do not pose a threat per se to aquatic organisms and plants because the toxic compounds are bound or encapsulated in the product. Substances that can be released unintentionally due to environmental influences during the life cycle of the perovskite solar cells containing lead or tin are to be evaluated as critical. These include above all heavy metals, acids and other substances that have not yet been identified. The level of toxicity of the pollutants depends on the solubility of the substances, but also on the pH of the environment (water, soil). Since these innovative perovskite solar cells are currently still in the research stage, there is only little data from the laboratory on the release quantities. Heavy metals themselves have already been well investigated toxicologically and existing limit values can be used in the future to better assess the risk to organisms from perovskite solar cells.
In an undamaged state, perovskite solar cells containing lead and tin do not pose a threat to environmental organisms. However, in the event of unwanted damage from environmental influences, degradation products can be released that have negative effects on organisms. According to current knowledge, replacing lead with tin in perovskite solar cells does not represent an alternative. Comparative studies with classic solar cells indicate that these too can release toxic substances. Perovskite solar cells based on lead or tin are currently (as of 2022) still part of the group of research materials and are not yet available in products on the market.
For new innovative solar cells, mainly perovskites containing lead or tin are currently being investigated. These heavy metal components, lead in particular, pose a real risk for humans especially during production and processing. Therefore, appropriate protective measures are necessary at the workplace.
Lead and tin are known toxic heavy metals. They can be taken up into the human body through inhalation or ingestion. They are distributed throughout the body via the blood system and can be deposited to a certain extent in the organs or ultimately in the skeleton (90% in the case of lead). Lead accumulated in bones, for example, has a half-life of up to 30 years. This means that after 30 years, about 50% of the accumulated lead has been degraded and removed from the bone. In contrast, about 95% of the tin absorbed is directly excreted from the body, and the remaining 5% have a half-life of several months in the various parts of the body. At the cellular level, the mechanisms of action of the two heavy metals are similar. Both lead and tin act as mimics of essential elements such as calcium, zinc or iron. They interfere with their natural function in enzymes, receptors or even the activity of red blood cells. Lead is classified as carcinogenic and has toxic effects on the genome, the immune system, the reproductive organs or embryonic development (see also lead poisoning).
Distribution and effects in the body
Up to now, lead containing perovskites for solar cells have only been tested in the laboratory on various cell lines. The investigated lung cells (A549, BEAS-2B), liver cells (HepG2) and intestinal cells (Caco-2) displayed different sensitivities to the tested perovskites and showed only minor damage after 24 h. It is likely that these observed minor damages are due to the released lead ions from the perovskite materials. However, further studies, especially on the underlying mechanisms, are needed for more accurate conclusions.
Lead is a known toxicant that can cause significant damage even in the smallest amounts. In acute or chronic lead poisoning, the heavy metal is deposited in the body and damages various organs and organ systems such as the digestive tract, the kidneys, the nervous system or even the bone marrow. For therapy, i.e. the removal of lead from the body, so-called chelating agents are used. These are able to bind the lead in the body to be then jointly excreted via the kidneys.
Lead or tin containing perovskites are particularly problematic for humans due to the heavy metals. The heavy metals lead and tin are known toxic substances that can be distributed and stored in the body after ingestion and cause various damages. Unintentional exposure and ingestion with these substances should be avoided at all costs. Currently, perovskite materials for solar cells are still in the research stage, so no products are yet available on the market (as of 2022).
Further Information
- Agency for toxic substances and disease registry (ASTDR) - Toxicological Profile for Lead (Last accessed: 09/2022)
- Agency for toxic substances and disease registry (ASTDR) - Toxicological Profile for Tin (Last accessed: 09/2022)
- Babayigit, A., A. Ethirajan, M. Muller, and B. Conings. "Toxicity of Organometal Halide Perovskite Solar Cells." Nat Mater 15, no. 3 (2016): 247-51. DOI:10.1038/nmat4572
- Dopp, Elke, and Albert W. Rettenmeier. "Tin, Toxicity." In Encyclopedia of Metalloproteins, edited by Robert H. Kretsinger, Vladimir N. Uversky and Eugene A. Permyakov, 2235-39. New York, NY: Springer New York, 2013. DOI:10.1007/978-1-4614-1533-6_118
Perovskite solar cells containing heavy metals can release various degradation products after damage from environmental influences (fire, storms, precipitation, floods). The lead and tin compounds that are primarily emitted can dissolve in water and form acids. The decomposition products can get into the air, water or soil. However, such perovskite solar cells containing lead or tin are still in the research stage (as of 2022) and are not available on the market as finished products.
Transport
Degradation products released from damaged lead or tin-containing perovskite solar cells can become airborne or be washed out of the solar cell by rain. Heavy metals such as lead or tin are released as ions and get either dissolved or as a solid in the soil or through rain events in surrounding waters. Since such innovative perovskite solar cells containing lead or tin are still in the research stage and are not used in practice, there are still no precise observations on the distribution of the decomposition products in the environment. Various approaches to encapsulation, in which the potentially toxic components are encased, offer significant protection against accidental release. This prevents critical substances from escaping if the solar cell is damaged by fire or water
Transformation
Heavy metals such as lead or tin are usually present in perovskite solar cells as organic heavy metal halides. If such solar cells are unintentionally damaged by environmental influences such as fire, hail or heavy rain, the built-in heavy metal compounds decompose and are released as heavy metal oxides or heavy metal halides. Distribution in the environment depends on many criteria, including soil conditions and the available amount of water in which the heavy metal compounds can be dissolved. The decomposition products of the perovskite solar cells containing lead or tin interact with substances present in the environment. Humic acids, for example, envelop released lead ions and thereby reduce their aggregation. This favors the transport of lead in the soil. A low pH value in the soil or water, on the other hand, favors the dissolution of the aggregates and the result is an increased release of lead ions. On the other hand, various positively charged ions in the soil have an aggregation-promoting effect on the lead aggregates and thus reduce their mobility in the soil.
Various degradation products such as heavy metals, heavy metal compounds and acids can be released from damaged perovskite solar cells containing lead or tin and spread into water and soil. Various environmental conditions such as the pH value, the presence of other substances and ions influence the transport and behavior of the released substances. However, such innovative perovskite solar cells are still in the research stage (as of 2022) and are not available as products on the market.
Further information
- Ren et al. 2021 (Review)
 >
>