>
Spotlight February 2022: Probabilistic risk assessment – the keystone for the future of toxicology
>
Spotlight February 2022: Probabilistic risk assessment – the keystone for the future of toxicology
The basics of toxicology are constantly being reconsidered, and the approach to risk assessment is therefore constantly being put to the test, because, as William Osler is cited in this publication, “Medicine (toxicology) is a science of uncertainty and an art of probability“.
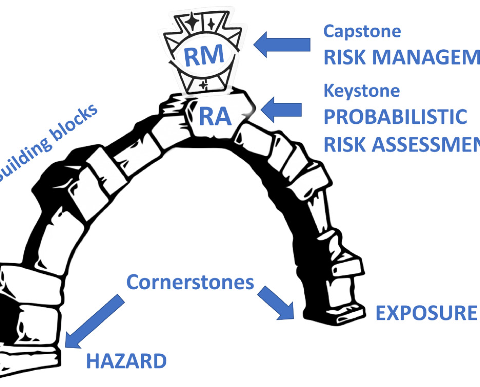
In this recent paper, the team around Thomas Hartung (Johns-Hopkins University/University of Konstanz) has shown that for improved toxicology we should rather work with a “Probabilistic Risk Assessment” approach. This is also or especially important for new materials, because with these there are particularly often gaps in knowledge, uncertainties in risk assessment due to conflicting data and the most diverse hypotheses and strategies of the various stakeholders. In the publication, various models are presented that are applicable for this type of risk assessment and for some of which corresponding software is also available to perform calculations for the respective exposure scenarios. In the examples for this approach, a paper by Jacobs et al. (1) is also cited here, who had applied the case to silica in food. They concluded that after taking all uncertainties into account and using all available data, the margin of safety has not yet been exceeded by far using silica in various food products. In 2017, an international group of experts applied this method to Titanium dioxide in seven different exposure scenarios and concluded no increased risk to humans, as the probability of exceeding the safety limits is vanishingly small (2).
The suggested approach by Johns Hopkins University is thus a good indication to adopt this method in order to be able to make a reasonable risk assessment for new, innovative materials even in the presence of uncertaintie.
Further literature:
- Jacobs, R., van der Voet, H., and Ter Braak, C.J. (2015). Integrated probabilistic risk assessment for nanoparticles: the case of nanosilica in food. J Nanopart Res 17, 251
- Tsang, M.P., Hristozov, D., Zabeo, A., Koivisto, A.J., Jensen, A.C.O., Jensen, K.A., Pang, C., Marcomini, A., and Sonnemann, G. (2017). Probabilistic risk assessment of emerging materials: case study of titanium dioxide nanoparticles. Nanotoxicology 11, 558-568
Original publication:
Maertens, A., Golden, E., Luechtefeld, T.H., Hoffmann, S., Tsaioun, K., and Hartung, T. (2022). Probabilistic risk assessment – the keystone for the future of toxicology. ALTEX 39, 3-29
Weitere Spotlights
Spotlight August 2020: The nanoGRAVUR Grouping approach
In August, we would like to present a paper of the German BMBF project nanoGRAVUR. nanoGRAVUR dealt from 2015-2018 with the grouping of nanostructured materials with regard to occupational safety, consumer and environmental protection and risk mitigation. The approach is now described by the project partners in this paper.Due to the variety of synthetic nanomaterials and the numerous modifications (differences in size, shape, chemical composition and surface functionalization), the effort required to investigate effects and behaviour within the framework of regulatory requirements is…
Read moreSpotlight June 2022: From small to clever – What does the future hold for the safety and sustainability of advanced materials?
The smallest particles in materials research, nanoparticles, have occupied us intensively for more than 20 years to elucidate and further investigate their safety for humans and the environment. Now, however, the development is going from “small = nano” to “clever = advanced”, as discussed in a contribution by international scientists. Thereby, it is a great […]
Read moreSpotlight March 2021: Is Nanotechnology the Swiss Army Knife against Future Pandemics?
The COVID 19 outbreak has led to a fundamental rethinking of existing approaches to diagnosis, treatment, and prevention methods. The need for better and more efficient concepts is global and urgent. Nanotechnology has long been at the forefront of innovation and has led to advances in many different disciplines. Could this interdisciplinary field help develop […]
Read moreSpotlight June 2023: New catalytic process for recovering important materials from composites in a single process
Previously virtually impossible and a huge problem: fibre-reinforced resin composites (epoxides) were not recyclable, and wind turbine rotor blades, for example, add up to a waste pile of 43 million tons by 2050. Researchers have now taken an important first step in “reprocessing” these composites and catalytically dissolving them so that the carbon fibres and […]
Read more