
Kaolin©RHJ-stock.adobe.com
Clays and especially nanoclays are used to improve the flame retardancy of plastics.
They are important components of soils and consist of layered silicates. Layered silicates are finely dispersed mineral particles based on the elements silicon, oxygen and other elements. Layered silicates are mined from natural sources and then mechanically and/or chemically treated. After further processing steps, they are used in various products. Due to their layered structure, they can swell when water accumulates between the layers or shrink when water is removed from them. They can increase in volume by up to sixfold through water absorption and form stable gels.
The global market for nanoclays is estimated by Transparency Market Research to grow to US$ 3.2 billion by 2023.
How can I come into contact with this material?
Clay minerals are ubiquitous in nature, we come into contact with them every day. However, the body is used to these amounts of clay minerals and so they do not pose a risk when present in normal amounts.
Since the nanoclay particles are embedded in plastic, direct human contact with significant quantities is unlikely. For food packaging, however, it has already been shown that they can also leach out.
Is there any risk from this material to humans and the environment?
Clay minerals can have both positive and negative influences in the environment and in humans. For positive influences, the clay minerals act like a sponge that filters toxins out of the environment. Negative influences occur when the clay mineral is already loaded with a large amount of toxins when it is taken up. Then toxins are released into the respective organism (human, animal or plant) over a longer period of time, and this can damage the organism.
In very large quantities, clay minerals, both in nanoform and as larger particles, can be harmful to humans and animals when inhaled; the body then cannot tolerate the large amount of dust. It has been shown for nanoclays: If these particles are inhaled in moderate quantities, then only minimal and temporary inflammation occurs in the lungs.
Conclusion
Clays/clay minerals, as well as nanoclays pose a low risk and only if they are present in high concentrations or loaded with toxins.
By the way….
Nanoclays are an example of naturally occurring nanomaterials.
Nanoclays are based on naturally occurring clays, which mainly consist of very fine mineral particles. The mineral particles of the clays mostly consist of layers of silicates, which are compounds based on the elements silicon, oxygen and other elements.
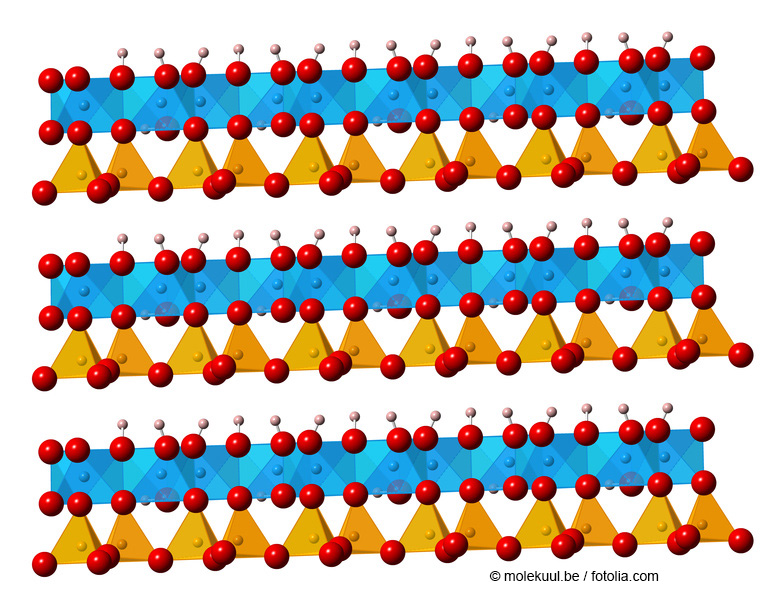
Molecular structure of Kaolin ©molekuul.be/fotolia.com
The basic building blocks of layered silicates are SiO4 tetrahedra. These building blocks can be connected to each other via common oxygen atoms. They can be thought of as "stacks" of 2-dimensional single, double or multiple layers. The layers are not linked to each other and can absorb or release water and other substances between the layers and can thus swell or shrink, which rigid framework silicates (see also zeolites) cannot do.
Layered silicates include groups of minerals such as mica, talc, kaolin, serpentine, montmorillonite or sepiolite. They differ, in one way, in the size and sequence of the areas in which the SiO4 tetrahedra are oriented upwards or downwards in the planes. Further, layered silicates can be differentiated by the presence of other elements, such as aluminium, that can take the position of the silicon components,.
Nanoclays are inflammable. They are used in flame and fire protection for plastics. They are added to the plastic, reducing the amount of flammable material. If the plastic catches fire, a separating layer is formed that inhibits it from burning further. The special properties of nanoclays, such as their chemical resistance, mechanical strength and thermal stability, have made them essential for today's electrical and electronics industry. Powder consisting of nanoscale nanoclays is also not self-igniting. Even as a finely dispersed mixture with air (dust) under the influence of an ignition source, nanoclays are not flammable, so there is no risk of a dust explosion.
Bentonites consist of nanoclays. Bentonitesare not a uniform substance but a mixture of different clay minerals. One main component is montmorillonite. Bentonites or montmorillonite are not a new material per se, but they are used in some innovative applications. A typical montmorillonite particle consists of layers about 1 nm thick which form large stacks. These large stacks contribute to the bentonite’s large total surface area, which makes the material a valuable absorbent. Absorption is a simple and effective strategy for the removal of pollutants or for water/wastewater treatment. An increasing number of publications on the absorption of toxic compounds by modified bentonites shows that there has recently been a growing interest in new low-cost absorbents for water treatment. Nanoclays can also bind radioactive caesium dissolved in water. Thus nanoclays can be used to reduce radioactive contamination like that from Fukushima .
In the animal feed industry, some bentonites have been approved by the German Ministry of Agriculture (BVL) to be used as an additive to bind fungal toxins, for example, and thus prevent them from causing harm.
Another use example is cat litter, which is typically composed of a mixture consisting of zeolites, sepiolites or bentonites or montmorillonites. This mixture is remarkable for its high absorption capacity for water and odour-causing components of cat urine and faeces.
When bentonite is mixed with water, the large stacks of montmorillonite adhere to each other and give the bentonite a strong binding capacity, making it suitable as a binder and as an additive to improve flow properties. Therefore, bentonite is often used as an ingredient in medicines and cosmetics and in industrial processes (e.g. for drilling fluids) to improve the rheology .
Other areas of application are, for example, as a plastic additive in food packaging films and plastic bottles. Furthermore, nanoclay-added plastics often have higher tensile strength, improved barrier and abrasion properties, excellent surface qualities, low thermal expansion and very good flow and processing properties. This outstanding performance profile makes plastic nanoclay composites an interesting alternative to conventionally reinforced materials. In addition, with the help of nanoclays, chemically incompatible plastics can be mixed to create a plastic that optimally combines the properties of both plastics. Recently, this has also been discussed for 3D printing applications, which means that plastic nanoclay composites could be regarded as an innovative material .
Natural occurrence and production

Montmorillon © Jean Paul Bounine / Fotolia.com
Nanoclays are mainly produced on the basis of the naturally occurring sedimentary rock bentonite, which in turn consists of 60 to 80 percent montmorillonite. Montmorillonite is named after a rock deposit near Montmorillon in southern France, bentonite after a clay deposit in Wyoming, USA near Fort Benton. Montmorillonite is an extra-fine clay mineral that is formed by the weathering of volcanic ash, tuffs and basic rocks (e.g. basalts). In Europe, important deposits are found, for example, on the Greek island of Milos and in Turkey and Germany. The largest German deposits are found in Bavaria.
Natural bentonites are preferred for many applications and products in the cosmetics and agricultural industries and also as cat litter.
In order to further increase the current growing capacity of natural bentonites in water for applications in the foundry industry, the construction and drilling industry and in the food sector, alkaline activation is often used. In this process, the cations (magnesium, aluminium, calcium) in the intermediate layers of the montmorillonite are replaced by sodium ions. In technical production, a kneading activation with soda (Na2CO3) is followed by a drying process, which is followed by a grinding step of the sodium montmorillonite.
Acid-activated bentonites are used in paper production and as catalysts. These activated Bentonites are used when large surfaces are required. Through reaction with acids, cations (magnesium, aluminium, iron) are extracted from the intermediate layers and replaced by hydrogen cations (protons). Depending on the degree of acid activation, large specific surfaces of up to 450 m²/g can be achieved .
Lipophilic bentonites are used to adjust the flow properties of oils, fats, ointments and plastics. These are preferably produced from alkali-activated bentonites and for this purpose are treated with organic nitrogen compounds (ammonium compounds), such as those found in commercially available fabric softeners. The organic modification makes the originally hydrophilic surface of the bentonite hydrophobic, thus allowing the complete dispersion of the individual layered silicate platelets in organic (hydrophobic) solvents. This is important if you want to use water-repellent nanoclays to specifically influence the flow properties of solvent-based paints and varnishes or to produce plastics.
Further Information
- TransparencyMarketResearch.com (EN):Nanoclay Market - Market for Packaging, Flame Retardants, Automotive, Paints & Coatings, and Other End-users - Global Industry Analysis, Size, Share, Growth, Trends and Forecast, 2015 - 2023 (https://www.transparencymarketresearch.com/nanoclays-market.html)
Clays in all particle sizes are contained in many products of daily life (fabric softeners, cosmetics, food packaging, cat litter) and thus we come into contact with them again and again. Particles from packaging could pass into food in small quantities. However, nanoclays can only cause health-relevant effects in extremely high concentrations.
Everyday Contact
Clays are used in many products because they have a very low impact on biological systems, i.e. they are not very toxic. For example, cat litter usually consists of several different bentonites, nanoclays are used in cosmetics, and for some years now it has also been allowed to be used in its nanoform in food plastic packaging in Europe to improve barrier properties. Nanoclays cannot enter the body through the skin and even if they are absorbed through the gastrointestinal tract, they tend to be harmless. Nanoclays can transfer from food packaging into food under certain circumstances. However, the amounts that can migrate out of the plastic are very small. They are below the approved limit of 10 mg/dm2 or cannot be detected at all .
However, the same applies to nanoclays as to other inert dusts, the dust should not be inhaled, if possible, at least not over a longer period of time or in high concentrations. Since some clay may also contain a significant amount of crystalline silicon dioxide (quartz), the regulations and limit values that also apply to quartz apply here.
Situation at the Workplace

Worker in the warehouse ©industrieblick/fotolia.com
In one of the world's largest production facilities for sepiolite, studies have shown that very high concentrations of these particles were released into the air, especially during packaging of the nanomaterials. This study has shown in 218 workers that massive exposure to sepiolite leads to lower lung function . As early as 1983, this was observed in Chinese workers in clay plants. Again, effects such as pulmonary fibrosis were found only at very high exposures . However, this very early work also showed that protective measures in workplaces were inadequate . In a more recent review, the authors summarize many available data. For example, bentonite alone is produced with a volume of more than 16 million tons (status 2014). For exposure, most data are only historical. From 1940 to 1990, occupational exposures of 1 - 92 mg/m3 (miners) and 20 - 40 mg/m3 (milling and packaging) were reported, and it was also pointed out that in recent times protective measures have been considerably improved, and thus the total occupational exposure is much lower than 30 - 60 years ago . For kaolin, the WHO defined in 2005 a specific form of pneumoconiosis, kaolinosis, which, however, appears only after very long exposure and is not critical. Kaolin has a measurable amount of crystalline silica (quartz), which is of great health concern when quartz particles are inhaled (formation of pulmonary fibrosis). However, the effect of kaolin is much weaker compared to quartz, yet there is a corresponding limit value for workplace exposure to kaolin.
Products and Consumer
Clays in their various manifestations, such as bentonite, kaolin, montmorillonite and others, as well as their modifications by surface alterations are ubiquitous. On the one hand, they are distributed in the earth's crust and thus contained in our natural environment and enter directly into the gastrointestinal tract when soil is swallowed (cf. geophagy or pica - eating earth). On the other hand, they are used in products of daily life (fabric softener, cat litter, cosmetics, packaging material and many more). Thus, we come into contact with them again and again.
Humans are exposed to clays in all particle sizes both as consumers and in the workplace. However, these have no adverse effects on humans or the environment, unless the quantities and concentrations are extremely high. Protective measures in the workplace help to avoid health effects. The quantities in products, on the other hand, are not problematic.
Further Information
- Nath, D., R, S., Pal, K., and Sarkar, P. (2022). Nanoclay-based active food packaging systems: A review. Food Packaging and Shelf Life 31
- European Commission (2011). Commission Regulation (EU) No 10/2011 of 14 January 2011 on plastic materials and articles intended to come into contact with food Text with EEA relevance. (last accessed: May 2022): https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=CELEX:32011R0010
- Adamis Z., Williams R.B., International Programme on Chemical Safety (2005). Bentonite, kaolin and selected clay minerals, ed. Geneva: World Health Organization, pp. 1-175, ISBN 9241572310
Particles are constantly being released from natural deposits into the air, soil and water as a result of environmental influences and are distributed there. For example, some of the suspended particles that make waters turbid are clay minerals. In applications in medicine, cosmetics, agriculture or the construction industry, release is likely, but has not yet been systematically investigated.
General release
Naturally occurring clay minerals are found all over the world. Not only do they represent the largest proportion of clays, but they are also indistinguishable from engineered nanoclays. The naturally occurring clay minerals are formed due to weathering of silicate-bearing rocks .

Kaolin mine ©Alexander Ruiz - stock.adobe.com
Technically engineered clays are released from waste in industry within the manufacturing process during the decomposition of polymers or in landfills, e.g. from coated food packaging. . Under certain conditions, particles of clay minerals can detach from larger clusters. Nanoclays were released from a plastic surface coated with clay minerals only by high temperatures (100 °C), without any mechanical stress taking place . However, there are studies that show that components of nanoclays found in packaging can dissolve and subsequently accumulate in packaged food . The resulting waste also makes it possible for nanoclays to enter the environment.
Released amounts
Due to the natural occurrence of clays, it is difficult to estimate or measure the release from products. Currently, no studies exist that have investigated the release from products (e.g., healing clay) or from specific industries (e.g., agricultural or construction industries).
Nanoclays are constantly released from natural sources. They either form rock layers at their place of origin or are transported away in the water and deposit at the bottom of lakes, rivers or seas. It is currently not possible to distinguish between engineered nanoclays and natural clay minerals.
Further information
- Bumbudsanpharoke, N & Ko, S (2015). Journal of Food Science Vol. 80, Nr. 5, 2015.
Humans and animals have a need to eat earth under certain circumstances. This behavior, also called pica or geophagy, sometimes serves to eliminate discomfort or other causes of physical discomfort and leads to the ingestion of clays. Nanoclays inhaled through the lungs can trigger inflammatory effects there.
Uptake via the Lung
As with all other poorly soluble dusts, nanoclays should not be inhaled in large quantities or for long periods of time. Nanoclays belong to the group of GBD particles (cf. cross-cutting text "Granular biopersistent dust particles"). In workplaces where workers were permanently exposed to high concentrations of these dusts, effects of inflammatory lung diseases and limitations of lung vital functions occurred more frequently . This was also confirmed in animal experiments with rats. The introduction of small nanofibers of sepiolite into the lungs of rats resulted only in temporary inflammation of the lungs. No negative effects on other organs were observed . Although these sepiolite nanofibers caused the formation of multinucleated giant cells in the lungs, all effects completely disappeared after several weeks.
Uptake via the Skin
Particles of various clay minerals are also used in cosmetics, for example in peelings as polishing particles. However, since these particles cannot enter the body through the skin, there are no studies on this, as there is no risk.
Uptake via the Gastro-Intestinal Tract
Nanoclays are found in toothpaste as polishing agents or in animal feed. If they are ingested in larger additions to food, they have no direct harmful effect. However, by binding essential substances in the food or the intestinal fluid, they can lead to an influence on food intake or to deficiency symptoms. The single intake of a larger quantity or frequent intake of small quantities of nanoclays usually has a negative effect in the organism only through contamination with trace elements .
The property to bind toxic metal ions (e.g. lead, cadmium) or naturally occurring carcinogenic fungal toxins (mycotoxins such as aflatoxin B1, fumonisin B1, etc.), makes them interesting as an additive to animal feed or for therapy for humans for detoxification. Aflatoxins may be present, for example, in contaminated corn used as feed for chickens. Studies in chicken farms showed that the addition of nanoclays reduced the toxicity of aflatoxins because the nanoclays bound the aflatoxin present in the feed . A similar protective effect was demonstrated in a study in which isolated human cells, as well as rats and humans (volunteers), were exposed to the fungal toxin fumonisin B1 . In all cases, the effect of the fungal toxin was significantly reduced. A comparable effect of montmorillonite could be demonstrated for other harmful substances, such as cadmium ions, when they were taken up together with the nanoclays via the gastrointestinal tract .
In addition to the positive effects of nanoclays, there are also repeated studies, mostly in vitro, which report possible damage to the organism. In an animal study, the extent to which nanoclays from plastic packaging could have negative effects in the organism was investigated. On two consecutive days, rats were administered large amounts of nanoclays (250, 500, and 1000 mg/kg body weight, respectively). Afterwards, the animals were examined for various parameters in the gastrointestinal tract and other organs. No uptake into the blood or other internal organs was observed. DNA damage was absent too. Hence, no toxicity could be demonstrated for nanoclays .
Uptake via medical application
The property of nanoclays to bind other chemical compounds or toxic elements makes them interesting for medical applications. For example, tests have been conducted to determine the extent to which nanoclays can be "filled" with drugs and then release the drugs after being absorbed by cells or an organism. Especially, anti-cancer drugs are of interest, as these have a damaging effect in the body and are supposed to be toxic only to cancer cells. Thus, attempts are being made to "package" these drugs in nanoclays in a way that is harmless to the body and then deliver them to the body. However, this way of application a cancer drug is still in the experimental stage .
The uptake of clays/nanoclays into the body cannot be ruled out both in the workplace (dust in the air) and for consumers (toothpaste, cat litter, food packaging). However, a health hazard is only expected if very large quantities are ingested or inhaled and is therefore unlikely in normal use.
Particles of clay minerals are ubiquitous and organisms are constantly exposed to them. In low concentrations they have no harmful effect. Clay minerals are good at binding various substances (metals, chemicals, drugs). This is exploited in many applications by using clay minerals as binders or carriers. Therefore, the effect of the substances bound to the clay minerals on environmental organisms is often considered.
Uptake in environmental organisms
Natural clay minerals are distributed everywhere. However, environmental organisms are frequently exposed to them only if they live either at the sites where clays are formed (in the form of rocks) or in waters in which flushed clay minerals are transported.
However, the uptake of clays in environmental organisms is always studied in combination with substances that bind well to clay minerals. In general, the uptake of the bound substance by the clay minerals can be reduced (i.e., the bound substance is no longer available to organisms) or enhanced (uptake together with clay mineral). Clay mineral particles provide a good surface for the accumulation of various nanoparticles. In the case of silver nanoparticles, accumulation on clay mineral particles can lead to enhanced exposure in aquatic organisms .
Clay minerals sampled from lakes show high enrichment in metals such as chromium, copper and nickel. The good adsorption of the metals is attributed to the surface properties of clay minerals. The surface has many reactive groups that react with the metals and bind them in this way. Thus, the clay minerals in the samples studied contribute to the removal of toxic metals from aquatic systems . However, the good binding of heavy metals also leads to sediments being heavily contaminated with them and disposal becoming necessary .
Toxicity in environmental organisms
Clay minerals consist of different layered silicates (e.g. kaolin or montmorillonite). The naturally occurring concentrations do not usually pose a hazard to environmental organisms. For example, kaolin has shown no toxic effects in studies with mosquito larvae. The clay mineral was even able to reduce the toxicity of an insect venom . Similar results were also shown for water fleas. Large concentrations of kaolin showed no negative effects. A possible explanation may be the high density of kaolin. Since kaolin sinks quickly in an aqueous environment and settles on the bottom, the exchange with the water fleas can only take place for a short time .
Toxicity of chemicals that bind to clay minerals
The toxicity of chemicals can be affected by binding to clays. In general, the toxicity of the bound substance can be reduced or increased in analogy to the uptake by the clay minerals.
Reduced toxicity
Clay minerals consist of different elements. A central component of clay minerals containing kaolin is aluminum. Such aluminum-containing clay minerals exhibit antibacterial properties in E.coli by damaging the cell membrane of the bacteria. This occurs due to the reaction of aluminum with the phospolipids in the cell membrane. Clay minerals can therefore play a possible role in combating multi-resistant germs .
Clay minerals have a promising role in medicine as a carrier material for drugs. Not only can they be loaded with a large amount of active ingredient (which is then released at the target site), but they are also heat-resistant and characterized by high tolerability. The antibacterial property of clay minerals often supports the effect of drugs .
The chemical-binding effect of clay minerals also affects earthworms. Toxic chemicals containing fluorine adsorb to clay minerals. Thus, the toxic compounds can no longer accumulate in the earthworms .
Increased toxicity
Clay minerals can also increase the effect of other chemicals or materials. This is mostly due to an increased uptake of the chemical in environmental organisms caused by the binding to the clay minerals.
Thus, particles of clay minerals form hetero agglomerates with silver nanoparticles. In experiments with zebrafish embryos, these agglomerates cause higher mortality than silver nanoparticles without clay mineral particle enrichment. Even at low concentrations, there was malformation of the tail and yolk .
The question of whether nanoparticles bound to clay minerals cause lower toxicity (due to the bound form) or higher toxicity (due to slow release over a longer period of time) has also been investigated in plant research. Zinc oxide nanoparticles bound to clay minerals have a more toxic effect in the plants "white mustard" and "Carex otrubae" than zinc oxide nanoparticles administered alone. The reason for this is that the bound zinc oxide nanoparticles or dissolved zinc ions can only be released slowly and the plants are exposed to zinc over a longer period of time .
Assessment of effects on environmental organisms
There are no specific toxicity values for naturally occurring clay minerals. They therefore do not generally pose a hazard to environmental organisms. However, particles bound to clay minerals can have a toxic effect. These are either toxic due to the combined effect of clay mineral and bound substance or pose a hazard due to the slow release of toxic substances.
Clay mineral particles are part of the natural habitat for environmental organisms. Even in high concentrations, no harmful effects occur. In applications, clay minerals often occur in combination with other substances, as they can bind them very effectively. For these combinations, both an weakening and an enhancing effect of clay minerals on the toxicity of the bound substances has been observed.
Since clays and nanoclays are found in a wide variety of applications, their possible effects in the organism are an important issue. When nanoclays are inhaled into the lungs as dust particles, they can cause inflammation. Via other uptake routes (ingestion or dermal) they have hardly any effects. In isolated cells, they have an effect only at very high concentrations. If organically modified, they can be effective at lower concentrations.
Distribution and Effects in the Body
Inhalation of dust can be hazardous to health. This also applies to dust particles from nanoclays. It has been shown both for humans and in animal experiments that large quantities of nanoclays in the lungs lead to inflammation. Exposure over two to four years to high concentrations of bentonite dust in the workplace (8 to 13 mg/m3) was shown to cause DNA damage in lung cells in Chinese workers. Although the statistics in this study are not sufficient, such an effect can certainly be assumed, since clay particles may have portions of crystalline quartz. Quartz also has such a DNA-damaging effect .
Already in 1993, similar effects were demonstrated in a study on Spanish workers. However, a different clay was processed here, sepiolite, a fibrous clay material . This can be critical because of its fibrous form (see cross-cutting text Airborne Fibres). The same material was tested on rats in animal experiments. There, a temporary inflammation occurred in the lungs, but this subsided after one week . Further effects on internal organs were not demonstrated. But so-called multinucleated giant cells were formed, which can be of importance in pneumoconiosis, among other things (compare Silicon Dioxide).
Further studies that introduced nanoclay particles into the lungs of mice and rats by instillation showed a comparable inflammation-inducing effect for bentonite. This was similar in magnitude to treatment with quartz. In contrast, kaolin was significantly weaker in its inflammatory effect .
If the occupational limit value for respirable dust in the workplace is observed, no health hazards to workers are to be expected.
Clays are also used as additives in animal feed. Therefore, the absorption and effect in the gastrointestinal tract was studied in mice and rats. Montmorillonite was added to the feed in large amounts (1000 mg/kg body weight in rats , and up to 5700 mg/kg body weight in mice in its pure form or after organic modification. Subsequently, it was investigated whether there could be inflammatory effects or the clay particles are absorbed into the body and affect other organs. In both studies, no damage or inflammation was observed, neither in the gastrointestinal tract itself nor in internal organs such as the liver or kidneys.
Although the clay particles have rather positive effects, e.g. as feed additives, it is certainly discussed that their property to bind specific ions or substances with a flat molecular structure leads to the animals showing deficiency symptoms in e.g. trace elements or vitamins . Thus, care must be taken to ensure that there is a balance in the feed between the added clays and the trace elements or micro-nutrients so that such deficiencies do not occur. However, the addition to the feed in Europe has been considerably restricted since May 2021, or the approvals in the EU register for feed additives have not been renewed in some cases.
Uptake and Effects in Cells
Toxicological studies on clays and nanoclays in cell cultures have been carried out mainly with two representative types: the bentonites, which include montmorillonite, and the kaolins, which include halloysite. Since these substances (mainly bentonite/montmorillonite) are approved as feed additives, but are also used in food packaging materials, it is important to understand their effect on the organism.
Halloysite was tested in liver and lung cells. Halloysite tubes between 200 nm and 1 micrometer in length showed little toxic effect and the cells responded only at high concentrations (50 - 100 µg/ml) and after three days of treatment. Lung cells are slightly more sensitive in this regard, but also only after several days of treatment with 20 and 40 µg/cm2 . The same group also investigated kaolin on another lung cell line. However, this was again much less sensitive and showed first cell toxic effects only at 100 µg/ml and above after five days of treatment. These studies indicate that kaolin has only a low toxic effect on cells ..
The results for bentonite or montmorillonite are comparable. The eight good-quality studies of the last two decades essentially state that pure bentonite/montmorillonite can hardly induce toxic effects in different cells. Skin cells, liver cells connective tissue cells, intestinal epithelial cells, Endothelzellen der Nabelschnur, umbilical cord endothelial cells and macrophages were studied. Only above 62.6 µg/ml or far beyond, the cells in the experiments showed a loss of their viability after one or more days of treatment. These concentrations are very high and are not reached in the workplace .
These clays especially nanoclays behave differently when they are chemically modified. The addition of nitrogen compounds (in this case quaternary ammonium compounds) changes the behavior and also the effect on cells. All studies that have compared pure montmorillonite with an organically modified variant come to the same conclusion, the organically modified nanoclay has a significantly more harmful effect on cells at lower concentrations. However, even with these variants, the concentrations are higher than would be expected in the workplace or even for consumers, so that no immediate risk to humans can be identified .
No effects of concern were found for clays and nanoclays in both animal and cell culture studies. Even though the organically modified nanoclays have an effect at lower concentrations compared to unmodified nanoclays, the concentrations used are much higher than in the workplace or in consumer products. A genotoxic effect is basically excluded, so that these substances are classified as safe by the European Food Saety Authority (EFSA).
Further information:
- European Commission, Directorate-General for Health and Food Safety (2021). European Union register of feed additives pursuant to Regulation (EC) No 1831/2003. Appendix 4(II), annex II, : List of additives subject to the provisions of Art. 10 § 2 of Reg. (EC) No 1831/2003 for which no application for reevaluation was submitted before the deadline of 8 November 2010 (Released 11.01.2021), Publications Office, 2021, https://data.europa.eu/doi/10.2875/0296
- EFSA Panel on Additives and Products or Substances used in Animal Feed (FEEDAP), Bampidis, V., Azimonti, G., Bastos, M.L., Christensen, H., Dusemund, B., Kouba, M., Kos Durjava, M., Lopez-Alonso, M., et al. (2019). Efficacy of a preparation of algae interspaced bentonite as a feed additive for all animal species. EFSA J 17, e05604. https://doi.org/10.2903/j.efsa.2019.5604.
- EFSA Panel on Additives and Products or Substances used in Animal Feed (FEEDAP), Rychen, G., Aquilina, G., Azimonti, G., Bampidis, V., Bastos, M.L., Bories, G., Chesson, A., Cocconcelli, P.S., et al. (2017). Safety and efficacy of bentonite as a feed additive for all animal species. EFSA J 15, e05096. https://doi.org/10.2903/j.efsa.2017.5096.
- EFSA Panel on Food Contact Materials, Enzymes, Flavourings and Processing Aids (CEF), Silano, V., Bolognesi, C., Chipman, K., Cravedi, J.P., Engel, K.H., Fowler, P., Franz, R., Grob, K., et al. (2018). Safety assessment of the active substances carboxymethylcellulose, acetylated distarch phosphate, bentonite, boric acid and aluminium sulfate, for use in active food contact materials. EFSA J 16, e05121. https://doi.org/10.2903/j.efsa.2018.5121
The surface of clay minerals can react well with various substances and microorganisms from the environment due to reactive groups. This property is used both for the removal of toxic substances from aqueous environments and for the application of catalysts.
Transport
Naturally occurring clay minerals are found in all environmental compartments and are transported back and forth between them. Clay minerals released from products cannot be precisely determined in terms of quantity, but probably account for a small proportion compared to the natural background.
Transformation
Clay minerals are durable materials that are mainly crushed by mechanical influences such as wind or water. Dissolution does not take place. Clay minerals have a very bonding-friendly surface and thus attach various substances. This effect is used in many applications. The properties and distribution in the environment are only slightly affected. Clays can bind chemicals, which also removes toxic nanoparticles from the environment. Titanium dioxide and silver nanoparticles aggregate with clays containing kaolin. However, the extent to which aggregation occurs depends on numerous factors such as pH, ionic strength or the presence of organic compounds (humic substance) . Drugs such as ibuprofen also bind to clays.
The good absorption capacity of clay minerals can also be used to filter drugs from sewage water. Montmorillonite-based clay minerals are very suitable for filtering beta-blocker drugs from water. However, the filtering ability depends on the temperature and pH value .
Clay minerals are also very good carriers for catalysts. Nanoscale catalysts can clump after a short time and their catalytic activity is reduced. However, if they are applied to surfaces of montmorillonite-based clay minerals, their activity is retained. Cobalt catalysts applied to clay minerals can thus degrade the antibiotic tetracycline, thereby reducing the risk to aquatic life .
Clay minerals occur in all environmental compartments and are predominantly transported in water and air. They are less mobile in soil. They effectively bind many other substances.
 >
>